- Blog
- Home
- New Page
- Gta San Andreas Download Full Game
- Windows 7 Premium Product Key Generator
- Doom Project Brutality 3.0 Download
- Woody Woodpecker Tomcat Combat
- Witcher 3 Graphic Mods
- Solidworks Free Download 2019 Free
- Arma 3 Breaking Point Chernarus Map
- Gta 5 Bodybuilder Mod
- Windows Live Calendar Update Error
- Witcher 3 Crashes To Desktop
- Baixar Avast Premier 2019 Crackeado
- Battle In The Forgotten City
- Space Battle Of Scarif
- War Is Hell Game
- Dnc Software For Fanuc Free Download
- Sim Settlements Far Harbor
- Focus Mk2 Remove Glovebox
- Install Hp Psc 950 On Windows 10
- Dbc File Viewer Online
- Chemical Structure Of Local Anaesthetics
- Pramukh Gujarati Font Converter
- A Deep Commitment To Purple
doi: 10.2344/0003-3006(2006)53[98:EOLAP]2.0.CO;2
PMID: 17175824
This article has been cited by other articles in PMC.
Abstract
It is impossible to provide effective dental care without the use of local anesthetics. This drug class has an impressive history of safety and efficacy, but all local anesthetics have the potential to produce significant toxicity if used carelessly. The purpose of this review is to update the practitioner on issues regarding the basic pharmacology and clinical use of local anesthetic formulations.
GENERAL PROPERTIES OF LOCAL ANESTHETICS
Local anesthetics interrupt neural conduction by inhibiting the influx of sodium ions. In most cases, this follows their diffusion through the neural membrane into the axoplasm, where they enter sodium channels and prevent them from assuming an active or “open” state. The local anesthetic molecule consists of 3 components: (a) lipophilic aromatic ring, (b) intermediate ester or amide chain, and (c) terminal amine. Each of these contributes distinct properties to the molecule (Figure 1).
Local anesthetic structure. All local anesthetics consist of 3 principal components, each contributing a distinct property.
The aromatic ring improves the lipid solubility of the compound, which can be enhanced further by aliphatic substitutions at locations designated (R). Greater lipid solubility enhances diffusion through nerve sheaths, as well as the neural membranes of individual axons comprising a nerve trunk. This property correlates with potency because a greater portion of an administered dose can enter neurons. Because bupivacaine is more lipid soluble than lidocaine, it is more potent and is prepared as a 0.5% concentration (5 mg/mL) rather than a 2% concentration (20 mg/mL).
The terminal amine may exist in a tertiary form (3 bonds) that is lipid soluble or as a quaternary form (4 bonds) that is positively charged and renders the molecule water soluble. As explained above, the aromatic ring determines the actual degree of lipid solubility, but the terminal amine acts as an “on-off switch” allowing the local anesthetic to exist in either lipid-soluble or water-soluble conformations. The tertiary and quaternary forms each play a pivotal role in the sequence of events leading to conduction block.
For the local anesthetic base to be stable in solution, it is formulated as a hydrochloride salt. As such, the molecules exist in a quaternary, water-soluble state at the time of injection. However, this form will not penetrate the neuron. The time for onset of local anesthesia is therefore predicated on the proportion of molecules that convert to the tertiary, lipid-soluble structure when exposed to physiologic pH (7.4). The ionization constant (pKa) for the anesthetic predicts the proportion of molecules that exists in each of these states. By definition, the pKa of a molecule represents the pH at which 50% of the molecules exist in the lipid-soluble tertiary form and 50% in the quaternary, water-soluble form. The pKa of all local anesthetics is >7.4 (physiologic pH), and therefore a greater proportion the molecules exists in the quaternary, water-soluble form when injected into tissue having normal pH of 7.4. Furthermore, the acidic environment associated with inflamed tissues favors the quaternary, water-soluble configuration even further. Presumably, this accounts for difficulty when attempting to anesthetize inflamed or infected tissues; fewer molecules exist as tertiary lipid-soluble forms that can penetrate nerves. In these situations, bupivacaine (pKa 8.1) would be least effective and mepivacaine (pKa 7.6) would be most likely to provide effective anesthesia (Figure 2).
Local anesthetic action. An injected local anesthetic exists in equilibrium as a quaternary salt (BH+) and tertiary base (B). The proportion of each is determined by the pKa of the anesthetic and the pH of the tissue. The lipid-soluble species (B) is essential for penetration of both the epineurium and neuronal membrane. Once the molecule reaches the axoplasm of the neuron, the amine gains a hydrogen ion, and this ionized, quaternary form (BH+) is responsible for the actual blockade of the sodium channel. Presumably, it binds within the sodium channel near the inner surface of the neuronal membrane.
The intermediate chain or linkage provides a convenient basis for classification and also determines the pattern of biotransformation. Esters are hydrolyzed by plasma esterases, whereas amides are biotransformed in the liver. Esters are no longer packaged in dental cartridges and are used infrequently with the exception of benzocaine, which is found in several topical anesthetic preparations.
Like other drugs, local anesthetics vary in their tendency to bind with plasma proteins. When circulating in the bloodstream, they bind to alpha-1-acid glycoprotein (acidic drugs more likely bind to albumin). This property of protein binding correlates with their affinity for protein within sodium channels and predicts the duration they will sustain neural blockade. Bupivacaine has the greatest percent protein binding and is the longest acting of local anesthetics available in dental cartridges. The clinical performance of local anesthetics correlates with 4 principal characteristics or properties that are summarized in Table 1.
Table 1
SYSTEMIC TOXICITY
Local anesthetics depress the central nervous system in a dose-dependent manner (see Figure 3). Low serum concentrations are used clinically for suppressing cardiac dysrhythmias and status seizures, but higher concentrations induce seizure activity. Convulsive seizures are the principal life-threatening consequence of local anesthetic overdose. Presumably this is due to selective depression of central inhibitory tracts, which allows excitatory tracts to run amuck. Evidence of lidocaine toxicity may commence at concentrations >5 μg/mL, but convulsive seizures generally require concentrations >8 μg/mL.
Systemic influences of lidocaine.
In addition to neural blockade, peripheral actions of most local anesthetics include varying degrees of vasodilation, and this contributes to the hypotension observed after administration of large doses. It is essential that local anesthetics be respected as central nervous system depressants, and they potentiate any respiratory depression associated with sedatives and opioids. Furthermore, serum concentrations required to produce seizures are lower if hypercarbia (elevated carbon dioxide) is present. This is the case when respiratory depression is produced by concurrent administration of sedatives and opioids. Goodson and Moore have documented catastrophic consequences of this drug interaction in pediatric patients receiving preoperative sedation, along with excessive doses of local anesthetics.
Before we address the variables that influence systemic serum concentrations, please consider the following suggestion for dose calculations. Percent solutions reflect grams of solute (drug) dissolved in 100 mL of solvent. A 3% mepivacaine solution contains 3 g mepivacaine dissolved in each 100 mL of solvent. Converting this to milligrams per milliliter, or milligrams per cartridge, is bothersome. For convenience, use the following suggestions. First of all, consider anesthetic cartridges as containing 2 mL, not 1.8 mL. This error will overestimate the dose and is therefore a safe practice. For a given percent solution, move the decimal 1 place to the right; the resulting number will reflect milligrams per milliliter. For example, 3.0% mepivacaine is 30 mg/mL. A dental cartridge contains 2 mL and therefore contains ~60 mg of mepivacaine. Bupivacaine 0.5% contains 5 mg/mL and therefore ~10 mg per cartridge. After injection of 2½ cartridges of 2% lidocaine it is convenient to consider this as ~5 mL at 20 mg/mL or 100 mg total.
In 1972, Scott et al published a study in a series of clinical studies assessing variables that determine subsequent concentrations of local anesthetics in serum. The serum concentration was found to vary according to the route by which the anesthetic is injected. Using lidocaine 400 mg, the highest serum levels illustrated in Figure 4 followed infiltration of vaginal mucosa and the lowest followed subcutaneous abdominal infiltration. In each case, however, peak serum level occurred 20–30 minutes after injection of lidocaine alone. Regardless of the route of administration, peak levels were reduced and the rate of absorption was delayed by adding vasopressors such as epinephrine to the local anesthetic solution. It is reasonable to assume that systemic concentrations after submucosal injection in the oral cavity would approximate those after injection into vaginal mucosa because of similar vascularity.
Serum concentrations following 3 routes of administration.
Additional variables were also addressed during these landmark studies. As expected, the dose and speed of injection are directly related to ultimate systemic serum concentration. A solution's concentration (eg, 2% vs 3%) is not relevant; the systemic concentration depends on the total dose (eg, 20 mL of 3% or 30 mL of 2% each amount to 600 mg and produce the same serum concentration). When using lidocaine or other anesthetics in concentrations >2%, one must consider the dose (milligrams) administered, not the volume (milliliters or cartridges).
Contrary to conventional thought, the age or weight of a patient does not predict systemic serum concentration following doses calculated as mg/y age or mg/kg. When managing pediatric patients, maximum doses are conventionally expressed in mg/kg, and this should be followed as a precaution. It is of little relevance for adults, however, and one should follow guidelines expressed as maximum dose in milligrams, regardless of weight or age. Obviously, this maximum amount should not be exceeded when calculating doses for large children.
When considering the toxicity of any drug class, one should be mindful of metabolites, as well as the parent drug. Local anesthetics are no exception. Lidocaine is biotransformed in the liver to monoethylglycinexylidide and glycinexylidide. These metabolites have significant activity and have been implicated in cases of lidocaine toxicity after repeated doses and continuous intravenous infusions.
A metabolite of prilocaine, 0-toluidine, can oxidize the iron in hemoglobin from ferrous (Fe2+) to ferric (Fe3+). Hemes so altered do not bind oxygen, and normal hemes on the same hemoglobin molecule do not readily release their oxygen. This form of hemoglobin is called methemoglobin, and when >1% of total hemoglobin is so altered, the condition is called methemoglobinemia. Patients appear cyanotic and become symptomatic when the proportion of methemoglobin exceeds 10 to 15%.3 The condition is rarely life threatening and responds to intravenous methylene blue, which reduces the hemes to their normal state. Methemoglobinemia attributed to prilocaine is unlikely to follow the administration of recommended doses. Rarely, one may encounter a patient with hereditary methemoglobinemia, which contraindicates the use of prilocaine.
ALLERGY TO LOCAL ANESTHETICS
It is not unusual for patients to claim they are allergic to local anesthetics. Upon careful questioning, however, one generally finds that what they experienced was either a syncopal episode associated with the injection or cardiac palpitations attributed to epinephrine either contained in the solution or released endogenously. Although rare, reports of allergic reactions to local anesthetics have appeared in scientific literature, but none of these have confirmed an IgE-mediated hypersensitivity reaction. Nevertheless, patients have occasionally experienced symptoms consistent with an allergic reaction to amide local anesthetics., In some cases, these episodes have been attributed to preservatives (methylparaben) or antioxidants (bisulfites) contained in the solution.6 Methylparaben is included in multidose vials to prevent microbial growth. It is no longer found in single-dose vials or dental cartridges. Metabisulfites prevent the oxidation of vasopressors and are included only in dental cartridges containing epinephrine or levonordefrin. To clarify several misconceptions regarding allergic reactions, a brief review of their pathogenesis is in order as presented by Adkinson.7
For drugs to be immunogenic, they must be of large molecular weight and possess multiple valences to be recognized by the immune cells. Large proteins such as insulin fulfill these requirements and are well established as immunogenic. Most other molecules are too small and must combine with other molecules that act as carriers, in which case the drug is described as a hapten. This complex of a carrier and hapten can induce and elicit an allergic reaction (Figure 5). Frequently, a metabolite of the drug is the actual molecule that functions as the hapten. This is true for beta lactam and sulfonamide antibiotics. In the case of sulfonamides, the phenyl ring containing an amine substitution is the moiety participating in the formation of the immunogenic complex (Figure 6). This moiety is common to other derivatives of para-aminobenzoic acid (PABA) such as methylparaben and some, but not all, ester local anesthetics. In these cases there may be the potential for cross allergenicity among similar compounds because of a common moiety (eg, sulfa antibiotics, methylparaben, and esters of PABA).
Carrier + hapten = immunogen.
Sulfonamide as hapten.
It is careless to describe “esters” as more allergenic than amide local anesthetics. An ester is merely a chemical linkage and imparts no immunogenicity to a compound. Rather, it is a molecular component joined by this linkage that is the culprit. This misconception has caused several agents to be inaccurately labeled as “cross-allergenic” with sulfonamides. Articaine is classified as an amide local anesthetic because of the linkage between its lipid-soluble ring and terminal amine. Its thiophene ring contains a sulfur atom, which has no immunogenic property, and an ester side chain that renders the compound inactive after hydrolysis. However, articaine does not liberate a metabolite resembling PABA and does not introduce concern regarding immunogenicity. In contrast, procaine is representative of esters derived from PABA and hydrolysis liberates a moiety that is potentially immunogenic (Figure 7).
Ester linkages of procaine and articaine.
A final misconception pertains to sulfites. These are included in local anesthetic solutions containing vasopressors to prevent their oxidation. They are inorganic compounds (−SO3) that have been implicated in allergic reactions, but they have no relation to immunogenicity attributed to PABA-related compounds. These agents are also used as antioxidants in fresh fruits and vegetables to preserve their color and overall appearance. Patients claiming allergy to such foods may experience cross-reactions with local anesthetic solutions containing vasopressors.
Drug reactions with clinical manifestations suggestive of immunological mechanisms, but known to lack an immune basis, are conventionally regarded as “pseudoallergic” reactions. Such reactions are idiosyncratic in mechanism and should be distinguished from true allergy. When these reactions mimic the more severe IgE-independent syndromes, they are described as “anaphylactoid” rather than anaphylactic. Anaphylactoid reactions involve the same final common pathway as true allergy, but the mechanism for the release of the vaso-active mediators is not immune mediated.
If a patient describes a reaction that is clinically consistent with allergy, the dentist should avoid using the offending agent until it is evaluated by an allergist. In the event an anesthetic is required before medical clearance can be obtained, the wisest choice would be either mepivacaine or prilocaine without vasopressors. Conventional wisdom holds that, if local anesthetics do indeed produce allergies, esters of PABA would be capable of cross-reacting, but this would not be likely among amide local anesthetics. Furthermore, by avoiding those solutions containing vasopressors, one avoids bisulfites that are included as antioxidants. Sensitivity to bisulfites is possible, especially among asthmatic or atopic patients. These principles are the basis for the flowchart presented in Figure 8. A patient should never be denied the benefit of local anesthesia because of flawed assumptions regarding allergy.
Managing patients allergic to local anesthetics. Rule out common reactions misinterpreted as allergy (eg, syncope and tachycardia). Then establish that the nature of their reaction at least resembled a hypersensitivity reaction (eg, rash, pruritus, urticaria, dyspnea). If the drug is known, choose another amide, free of vasopressor so no bisulfites are present. Otherwise, refer to an allergist, sharing this figure if necessary. Adapted from deShazo RD, Kemp SF. JAMA. 1997;278:1903.
LOCAL ANESTHETIC COMPARISONS
Cocaine was the first local anesthetic, discovered in 1860. It is unique among the local anesthetics because, in addition to blocking impulse conduction along axons, it inhibits reuptake of neurotransmitters by adrenergic neuronal endings. Peripherally, this results in an accumulation of norepinephrine within sympathetic synapses leading to vasoconstriction and cardiac stimulation. Centrally, accumulation of norepinephrine results in generalized central nervous system stimulation. However, additional adrenergic neurotransmitters are found within the central nervous system, and their reuptake is inhibited as well. In particular, cocaine's inhibition of dopamine reuptake is pronounced and responsible for its euphoric effect and potential for abuse. For all these reasons, and because it has no greater anesthetic efficacy, cocaine receives little use medically except for topical application during certain ear-nose-throat and ophthalmologic procedures. Because of the potential for added sympathomimetic effects, epinephrine should be avoided when administering local anesthetics to patients suspected of recent cocaine consumption.
Procaine (Novocain) was introduced in 1905 and became the first local anesthetic to gain wide acceptance in the United States. However, its popularity declined after the introduction of lidocaine in 1948. Today, lidocaine is the most widely used agent, but all local anesthetics have comparable efficacy. They differ in potency and several pharmacokinetic parameters that account for differences in the onset and duration of anesthesia. Selection of a particular agent must take into account the duration of the procedure planned and issues regarding vasopressor concentrations. For lengthy procedures, bupivacaine is the logical choice, but it has been implicated as one of the more painful agents during injection according to studies that have compared various anesthetics. One strategy is to provide the initial 60–90 minutes of anesthesia with a less irritating agent (lidocaine or prilocaine) and then reinject the anesthetized tissue with bupivacaine to provide analgesia well into the postoperative period. Such a strategy is most effective after nerve blocks; shorter duration should be anticipated after soft tissue infiltration. See Table 2 for durations and maximum doses.
Table 2
Despite anecdotal claims regarding the superiority of articaine (Septocaine, Ultracaine), double-blind studies have confirmed that efficacy is comparable with lidocaine., Nevertheless, articaine has certain features that make it an attractive choice for selected cases. Unlike other anesthetics having benzene as their aromatic ring, articaine has a thiophene ring, which confers greater lipid solubility than lidocaine. This property should have allowed a lower concentration, but in fact it was formulated as a 4% solution. Claims for successful anesthesia after infiltration of the mandible are likely attributable to high lipid solubility and more molecules per milliliter injected when compared with lidocaine. To date, there have been no published studies comparing articaine with 4% lidocaine solutions for mandibular infiltration. Furthermore, such concentrations of lidocaine present an unacceptable risk for systemic toxicity, and this concern introduces another attractive property of articaine, namely, pattern of clearance.
Although articaine is classified as an amide because of linkage of its intermediate chain, the thiophene ring also contains an ester side chain. This chain is hydrolyzed by plasma esterases rendering the molecule inactive. The result is that articaine has a half-life of only 20 minutes compared with ~90 minutes for lidocaine and other amides that require hepatic clearance. For this reason, articaine presents less risk for systemic toxicity at equipotent doses (eg, 1 cartridge 4% articaine vs 2 cartridges 2% lidocaine). Presumably, this principle would also confer greater safety during lengthy appointments when additional doses of anesthetic must be administered. It should be clarified that articaine is classified molecularly as an amide, not an ester of PABA, and does not present any concern for cross-allergy to PABA derivatives.
The advantages of articaine are tempered somewhat by reports of paresthesia after its use for inferior alveolar blocks. Haas and Lennon reported an increased incidence of paresthesias in Canada after the introduction of articaine in the mid 1980s. In 1993 alone, 14 cases of paresthesia were reported, and all were attributed to articaine or prilocaine (see Table 3). Although the overall incidence of paresthesia is low, one cannot discount the increased risk that is apparently associated with higher concentrations of local anesthetics when used for nerve blocks.
Table 3
As part of the approval process for a new drug in the United States, certain data must be submitted to the US Food and Drug Administration. When articaine was being submitted for approval, the following data were part of that application process and are consistent with those of Haas and Lennon. The entire text of the document may be found by searching the US Food and Drug Administration web site for the Statistical Review of Application Number 20-971. Looking at study #96001.02US on page 12 of the document, it reads, “Once again, the articaine group appears to have significantly higher risk of paresthesia (10 in 569, vs. 1 in 284 in the lidocaine group).” So, it appears there is a concern with respect to paresthesia when 4% local anesthetic solutions are used. As with all drugs, each practitioner needs to perform a “risk-benefit” analysis before using a medication. Only if the benefit of using articaine outweighs the risk for this practitioner in this patient should it be considered for use. It might be wise to limit the use of these agents for infiltration and reserve their use in nerve blocks for failed attempts with other agents.
MAXIMUM DOSES FOR LOCAL ANESTHETICS
The final issue to be considered is the maximum amount of anesthetic that can be used in a given patient. According to the data originally presented by Scott et al, lidocaine 400 mg injected submucosally produces systemic serum concentrations well below toxic levels. This is approximately the amount found in 10 anesthetic cartridges, and this number is conventionally cited as the limit per dental appointment. Summaries of these studies, and others regarding additional local anesthetics, are the basis for the maximum recommended doses listed in Table 2. But current information has not addressed issues regarding lengthy dental procedures that may well outlast the duration of anesthesia provided by conventional agents. In such cases, decisions regarding additional doses must be predicated on empiric judgment.
The serum half-life (T1/2) of the various local anesthetics ranges from 90 minutes for conventional agents such as lidocaine to nearly 300 minutes for agents such as bupivacaine. This decline commences after peak concentration is achieved and declines after approximately 20–30 minutes with anesthetics alone. (The time to peak concentration when combined with vasopressors is not well documented, but adding an additional 10–15 minutes would be a reasonable estimate.) Once the peak concentration is achieved, additional doses will absorb as original doses are in decline. This is a perilous time because one cannot accurately predict the serum concentration at any period. Additionally, individual patient values cannot be absolutely known because of the bell-shaped pattern of distribution for patient responses and renders these theoretical calculations even more problematic. However, given the fact that solutions containing vasopressors are absorbed slowly and their peak concentrations are reduced, it seems reasonable to assume that doses that do not exceed one fourth of the maximum permissible dose listed in Table 2 could be administered during each subsequent hour of treatment. This suggestion presumes the patient has adequate hepatic perfusion and function and is not medicated with agents that delay hepatic clearance. During lengthy procedures, however, one should anesthetize and complete treatment in one region before anesthetizing another. By following this principle, it is rarely necessary to exceed maximum recommended limits. When using a combination of agents, guidelines for maximum doses should be regarded as additive. For example, if half the maximum dose for lidocaine has been administered, it would be safe to administer up to half the maximum dose for mepivacaine.
VASOPRESSORS
Vasopressors are combined with local anesthetics to provide local hemostasis in the operative field and to delay their absorption. Delayed absorption of local anesthetics not only reduces the risk for systemic toxicity, but also prolongs the duration of anesthesia. Epinephrine is the most common agent used for this purpose, despite the fact that it exhibits considerable cardiac stimulation because of its beta-1 agonistic action in addition to its desired vasoconstrictive activity (alpha-1 agonistic action).
Despite the popularity of epinephrine 1 : 100,000, concentrations >1 : 200,000 (5 μg/mL) offer no advantage in terms of prolonging anesthesia or reducing serum concentrations of local anesthesia. Higher concentrations do not provide better onset or duration for inferior alveolar nerve block, or reduce local anesthetic serum concentrations. However, greater concentrations (eg, 1 : 100,000 [10 μg/mL] and 1 : 50,000 [20 μg/mL]) may provide better hemostasis at the surgical site when this influence is desired.
To properly address safety issues regarding vasopressors, one must first appreciate principles of dose calculations. Solutions expressed as ratios represent grams of solute (drug) dissolved in milliliters of solvent. A 1 : 100,000 concentration of epinephrine contains 1 g epinephrine dissolved in 100,000 mL solvent. Converting this to milligrams or micrograms per milliliter is time consuming. This concentration is most common in local anesthetic solutions and is best memorized as 10 μg/mL. If one accepts the earlier suggestion that cartridges contain 2 mL, it follows that a cartridge of local anesthetic combined with epinephrine 1 : 100,000 contains ~20 μg epinephrine. Simply double this amount when using solutions that contain epinephrine 1 : 50,000, and halve those containing epinephrine in concentrations of 1 : 200,000.
There is continued debate regarding deleterious influences of vasopressors on patients with cardiovascular disease. Clinical trials have confirmed unequivocally that even small doses of epinephrine in local anesthetic solutions have an influence on cardiovascular function (see Figure 9). Dionne et al studied the influence of 3 cartridges of 2% lidocaine with epinephrine 1 : 100,000 (~60 μg or precisely 54 μg epinephrine). Submucosal injection of this dose increased cardiac output, heart rate, and stroke volume. Peripheral resistance was reduced, and mean arterial pressure remained essentially unchanged. This finding was consistent with that according to 10 μg/min epinephrine infusions presented in standard texts.18 The issue we must address is whether these influences pose a significant risk to patients with cardiovascular diseases. Putative standards and guidelines continue to be presented but in fact are all anecdotal. Ultimately, the decision requires the dentist to exercise sound clinical judgment based on a thorough analysis of each patient under consideration. If consultation with the patient's physician is indicated, discuss the anticipated dose range in terms of micrograms, not concentrations or cartridges. For example, if 2–4 cartridges of local anesthetic are planned, explain that you will be using 40–80 μg of epinephrine infiltrated sub-mucosally, not 2–4 cartridges of epinephrine 1 : 100,000. The physician is unfamiliar with a dose expressed as cartridges or concentrations. As reference, consider the conventional epinephrine dose for allergic reactions is 0.3 mg or 300 μg.
Cardiovascular influences of epinephrine. Patients received submucosal infiltration of 3 cartridges (5.4 mL) of 2% lidocaine and 2% lidocaine with epinephrine 1 : 100,000. Changes in cardiovascular parameters were recorded as percent change. Heart rate (HR), stroke volume (SV), and cardiac output (CO) determine systolic blood pressure. Peripheral resistance (PR) determines diastolic blood pressure. Mean arterial pressure (MAP) is calculated as (SBP + 2 × DBP)/3. Adapted from Dionne et al.
Levonordefrin (NeoCobefrin) is the vasopressor combined with 2% mepivacaine solutions in the United States. It closely resembles norepinephrine rather than epinephrine and lacks activity at Beta2 receptors. Epinephrine increases heart rate and systolic pressure but lowers diastolic pressure. In contrast, systemic administration of norepinephrine increases systolic, diastolic, and mean arterial pressures, and this triggers a reflex slowing of heart rate.18 (This is illustrated and explained in Figure 10.) Levonordefrin has been suggested as an alternative to epinephrine-containing local anesthetics when treating patients with cardiovascular heart disease because it does not increase heart rate. However, advocates fail to consider its undesirable influence on blood pressure. The 1 : 20,000 levonordefrin concentration found in mepivacaine is considered equipotent to standard 1 : 100,000 epinephrine concentrations in terms of alpha receptor activity (vasoconstriction). After infiltration, they have equivalent efficacy for constricting submucosal vessels, and their influences on local hemorrhage and anesthetic absorption are similar.
Cardiovascular influences of norepinephrine (and levonordefrin) versus epinephrine.18 A. Both drugs stimulate Beta1 receptors on cardiac muscle, which increase myocardial contractility. This results in an increase in systolic pressure. B. Both drugs stimulate alpha receptors on vessels, which causes them to constrict. Submucosal vessels contain only alpha receptors, so both drugs produce local vasoconstriction when injected submucosally. But submucosal vessels are not illustrated here; they do not influence diastolic pressure. Systemic arteries influence diastolic pressure and contain Beta2 receptors, which vasodilate and are far more numerous than alpha receptors. Norepinephrine has no affinity for Beta2 receptors and constricts systemic arteries by activating the alpha receptors, even though they are less numerous. This increases diastolic pressure. Epinephrine, which has Beta2 as well as alpha receptor activity, produces vasodilation and a reduction in diastolic pressure. C. Both drugs stimulate Beta1 receptors on the Sino-atrial node, which increases heart rate. But this potential effect from norepinephrine is overridden by a reflex explained as follows. Notice that epinephrine has no influence on mean arterial pressure; systolic pressure increases but diastolic decreases and negates any effect on mean arterial pressure. Norepinephrine increases systolic, diastolic, and mean arterial pressures. The increase in mean arterial pressure stimulates baroreceptors in the carotid sinus, which trigger a vagal slowing of heart rate.
Misconceptions regarding adverse interactions between vasopressors and antidepressants persist despite literature that dispels this concern. Most of these concerns relate to pharmacokinetic interactions predicated on clearance of catecholamines. Although neuronal uptake is the principal method for termination of endogenous adrenergic neurotransmitters, hepatic biotrans-formation is the principal pathway for termination of exogenously administered adrenergic drugs.18 Most noncatecholamines are metabolized in liver by mono-amine oxidase, but those having a catecholamine structure are primarily inactivated by catechol-o-methyltransferase. Epinephrine and levonordefrin are catechol-amines and are metabolized primarily by catechol-o-methyltransferase, not monoamine oxidase or neuronal uptake.18,19 Patients treated with monoamine oxidase inhibitors are not hindered in clearing either of these vasopressors.11 Newer antidepressants (eg, fluoxetine [Prozac]) selectively inhibit serotonin reuptake and likewise are not a concern. Although they are not contra-indicated entirely, vasopressors should nevertheless be used with caution in patients receiving tricyclic antidepressants. This particular class of antidepressants has an ability to produce cardiac arrhythmias, which could be potentiated by a similar tendency shared by all sympathomimetic agents. For these patients, vital signs should be monitored continually if more than 1 or 2 cartridges containing any vasopressor are used.
Patients medicated with nonselective beta-blocking agents have heightened sensitivity to the systemic effects of vasopressors. Beta blockers are used for their ability to block sympathetic influences on cardiac Beta1 receptors. Unfortunately, older agents are nonselective and also block Beta2 receptors on systemic arteries. This offsets the tendency of epinephrine to dilate and instead increases systemic vascular resistance. Blood pressure increases dramatically and is followed by a reflex slowing of heart rate. The mechanism of this interaction is detailed in Figure 11 and Table 4. Numerous reports of stroke and cardiac arrest have been reported in medical literature warning of this potential interaction., Hypertensive responses have been reported after low doses of both epinephrine and levonordefrin. It is significant that this interaction will not occur in those patients receiving selective beta1 antagonists because, at conventional doses, these have little or no affinity for vascular beta2 receptors. However, caution is still advised because putative selectivity for beta1 receptors may not be absolute, especially in patients medicated with high doses. The following are 2 guidelines to consider when using vasopressors in patients medicated with beta blockers:
- Avoid the use of vasopressors, if reasonable.
- If a vasopressor is to be used, record blood pressure and heart rate, then proceed as follows: (a) after the injection of each cartridge, pause 5 minutes and reassess vital signs before administering any additional local anesthetic; or (b) infiltrate the entire region to be treated by using a cartridge to provide constriction of local vessels, then reinject the region with a local anesthetic free of vasopressor.
Mechanism of epinephrine–beta-blocker interaction. The cardiovascular influences of epinephrine are mediated via alpha, Beta-1, and Beta-2 receptors and are altered in patients medicated with nonselective beta blockers. These graphs distinguish typical cardiovascular responses after a 10-to 20-μg test dose of epinephrine in a normal versus a beta-blocked patient (see Table 4 for additional explanation). Note that the key underlying mechanism involves the influence of epinephrine on systemic vascular resistance and subsequent diastolic blood pressure (DBP).
Table 4
Mechanism of Epinephrine/Non-selective Beta-Blocker Interaction (see Figure 11 for illustration)
Finally, some consideration should be given to maximum permissible doses of vasopressors. To express limits in terms of appointments is impractical; time of treatment may be as brief as 30 minutes or as long as 3–4 hours. The influence of a given dose of epinephrine among patients is highly variable. Peak serum levels of epinephrine after submucosal injection are generally achieved within 5 minutes and decline rapidly due to inactivation by catechol-o-methyltransferase. Generally, the hemodynamic influences of epinephrine are witnessed within minutes of injection and have completely subsided in 20 minutes. A dose of 40 μg (approximately 2 cartridges containing epinephrine 1 : 100,000) is the most conservative and frequently cited dose limitation for epinephrine in patients with significant cardiovascular disease. It should be clarified that this guideline more accurately reflects 30-minute time periods, not appointments. A more reasonable suggestion should be based on patient assessment, not maximal dose. If for any reason the medical status of a patient is in question, reassess vital signs within 5 minutes after the administration of each cartridge. If the patient is stable, additional doses may be administered followed by a similar pattern of reassessment.
CONTINUING EDUCATION QUESTIONS
- Beckercaine is a newly released local anesthetic classified as an ester and is compounded with epinephrine 1 : 200,000. It has twice the lipid solubility of lidocaine, and other data are as follows, with corresponding data for lidocaine in parentheses: pKa = 7.5 (7.9) Protein binding 92% (65%). All of the following are correct regarding this new wonder-drug EXCEPT:
- It is likely formulated as a 1% solution.
- It has a faster onset than lidocaine.
- It is metabolized in plasma.
- It has a shorter duration than lidocaine.
- Which of the following reflect accurate doses contained in 5 mL (~2.5 cartridges) prilocaine 4% with 1 : 200,000 epinephrine?
- Prilocaine 20 mg; epinephrine 50 μg.
- Prilocaine 200 mg; epinephrine 25 μg.
- Prilocaine 20 mg; epinephrine 25 μg.
- Prilocaine 200 mg; epinephrine 10 μg.
- Hypertensive events attributed to drug interaction have occurred after the administration of local anesthetics containing epinephrine in patients medicated with which of the following?
- Nonselective beta blockers.
- MAOI antidepressants.
- SSRI antidepressants.
- a and b are correct.
- a, b, and c are correct.
- The maximum recommended dose is 500 mg for lidocaine with epinephrine and 400 mg for mepivacaine. Anesthesia is difficult to obtain, and you have administered 6 cartridges of 2% lidocaine with epinephrine to remove 4 third molars. Two sites remain sensitive, and you elect to reinject with 3% mepivacaine to avoid more epinephrine. Which of the following number of cartridges would be the maximum number you can safely administer? (Assume 2 mL per cartridge.)
- 1
- 3
- 5
- 7
REFERENCES
- Goodson JM, Moore PA. Life-threatening reactions after pedodontic sedation: an assessment of narcotic, local anesthetic and antiemetic drug interactions. J Am Dent Assoc. 1983;107:239–245. [PubMed] [Google Scholar]
- Scott DB, Jebson PJR, Braid DP, et al. Factors affecting plasma levels of lignocaine and prilocaine. Br J Anaesth. 1972;44:1040–1049. [PubMed] [Google Scholar]
- Prchal JT, Gregg X. Hemoglobinopathies: methemoblobinemias, polycythemias and unstable hemoglobins. In: Goldman L, Ausiello D, editors. Cecil Textbook of Medicine. 22nd ed. Philadelphia, Pa: WB Saunders Co; 2004. [Google Scholar]
- Gall H, Kaufmann R, Kalveram CM. Adverse reactions to local anesthetics: analysis of 197 cases. J Allergy Clin Immunol. 1996;97:933–937. [PubMed] [Google Scholar]
- Berkun Y, Ben-Zvi A, Levy Y, Galili D, Shalit M. Evaluation of adverse reactions to local anesthetics: experience with 236 patients. Ann Allergy Asthma Immunol. 2003;91:342–345. [PubMed] [Google Scholar]
- Schatz M. Adverse reactions to local anesthetics. Immunol Allergy Clin North Am. 1992;12:585–609.[Google Scholar]
- Adkinson NF., Jr . Drug allergy. In: Adkinson NF Jr, Yunginger JW, Busse WW, editors. Middleton’s Allergy: Principles and Practice. 6th ed. Philadelphia, Pa: Mosby Inc; 2003. et al, eds. [Google Scholar]
- Morris R, McKay W, Mushlin P. Comparison of pain associated with intradermal and subcutaneous infiltration with various local anesthetic solutions. Anesth Analg. 1987;66:1180–1182. [PubMed] [Google Scholar]
- Wahl MJ, Overton D, Howell J, Siegel E, Schmitt MM, Muldoon M. Pain on injection of prilocaine plain vs. lidocaine with epinephrine. A prospective double-blind study. J Am Dent Assoc. 2001;132:1396–1401. [PubMed] [Google Scholar]
- Wahl MJ, Schmitt MM, Overton DA, Gordon MK. Injection pain of bupivacaine with epinephrine vs. prilocaine plain. J Am Dent Assoc. 2002;133:1652–1656. [PubMed] [Google Scholar]
- Yagiela JA. Local anesthetics. In: Dionne RA, Phero JP, Becker DE, editors. Management of Pain & Anxiety in the Dental Office. St Louis, Mo: WB Saunders/Elsevier Science; 2002. [Google Scholar]
- Malamed SF, Gagnon S, Leblanc D. Efficacy of articaine: a new amide local anesthetic. J Am Dent Assoc. 2000;131:635–642. [PubMed] [Google Scholar]
- Mikesell P, Nusstein J, Reader A, Beck M, Weaver J. A comparison of articaine and lidocaine for inferior alveolar nerve blocks. J Endod. 2005;31:265–270. [PubMed] [Google Scholar]
- Haas DA, Lennon D. A 21 year retrospective study of reports of paresthesia following local anesthetic administration. J Can Dent Assoc. 1995;61:319–330. [PubMed] [Google Scholar]
- Dagher FB, Yared GM, Machtou P. An evaluation of 2% lidocaine with different concentrations of epinephrine for inferior alveolar nerve block. J Endod. 1997;23:178–180. [PubMed] [Google Scholar]
- Tofoli GR, Ramacciato JC, de Oliveira PC. Comparison of effectiveness of 4% articaine associated with 1: 100,000 or 1:200,000 epinephrine in inferior alveolar nerve block. Anesth Prog. 2003;50:164–168. et al. [PMC free article] [PubMed] [Google Scholar]
- Dionne RA, Goldstein DS, Wirdzek PR. Effects of diazepam premedication and epinephrine-containing local anesthetic on cardiovascular and catecholamine responses to oral surgery. Anesth Analg. 1984;63:640–646. [PubMed] [Google Scholar]
- Hoffman BB. Catecholamines, sympathomimetic drugs, and adrenergic receptor antagonists. In: Hardman JG, Limbird LE, Gilman AG, editors. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill; 2001. [Google Scholar]
- Lawson NW, Meyer DJ. Autonomic nervous system physiology and pharmacology. In: Barash PG, Cullen BF, Stoelting RK, editors. Clinical Anesthesia. 3rd ed. Philadelphia, Pa: JB Lippincott Co; 1997. [Google Scholar]
- Becker DE. Clinical implications of autonomic pharmacology. J Oral Maxillofac Surg. 1992;50:734–740. [PubMed] [Google Scholar]
- Foster CA, Aston SJ. Propranolol-epinephrine interaction: a potential disaster. Plast Reconstr Surg. 1983;72:74–78. [PubMed] [Google Scholar]
- Gandy W. Severe epinephrine-propranolol interaction. Ann Emerg Med. 1989;18:98–99. [PubMed] [Google Scholar]
- Mito RS, Yagiela JA. Hypertensive response to levonordefrin in a patient receiving propranolol: report of a case. J Am Dent Assoc. 1988;116:55–57. [PubMed] [Google Scholar]
Articles from Anesthesia Progress are provided here courtesy of American Dental Society of Anesthesiology
Abstract
Br J Anaesth 2002; 89: 52–61
Local anaesthetics have been used clinically for well over a century, but the molecular mechanisms by which they alter specific functions of the peripheral nerve system remained unclear for a long time. Investigations during the last few decades presented evidence implicating the sodium (Na+) channel protein as a target for specific, clinically important, local anaesthetic effects on mammalian neurones. In this review I will present the state of knowledge of the action of local anaesthetics at the molecular level, with an emphasis on Na+ channels. After a brief survey of the history of concepts of local anaesthetic actions, the structure of the Na+ channel will be described with regard to its function in order to understand the recent findings that have followed the description of the amino acid sequence of the Na+ channel protein.46 At clinically relevant concentrations local anaesthetics can also affect potassium (K+) and calcium (Ca2+) channels, which might help explain some of their side‐effects. General anaesthetics also interact with these targets (ion channels), but differences in selectivity compared with local anaesthetics have been reported. Current findings indicate that local anaesthetics also act on intracellular mechanisms, which raises the question of whether these might explain toxicity and other side‐effects.
Historical view
Cocaine was first used as a local anaesthetic by Carl Koller and Sigmund Freud (the latter the founder of psychoanalysis). They noticed a numbing effect on the tongue after swallowing cocaine, and Koller, who was intent on finding a drug to anaesthetize the cornea, knew that Freud had relieved pain with cocaine.26 In fact they could demonstrate on themselves—using pins to touch their cornea in front of a mirror—that within minutes of applying cocaine they could no longer feel touch or pain. In 1884 they reported that they had enucleated painlessly a dog’s eye. Leonard Corning, a neurologist in New York City, had tried in 1885 to inject cocaine solution (2%) between the spinous processes in a young dog, which resulted in insensibility within 5 min;19 this procedure was subsequently tried on patients, with the drug presumably acting in the epidural space. The lumbar puncture later introduced by Quincke produced spinal anaesthesia.54 Cocaine was widely used despite its disadvantages of high toxicity, short duration of anaesthesia, impossibility of sterilizing the solution and its cost (not to mention addiction). When in 1904 Alfred Einhorn, investigating degradation products of cocaine, synthesized procaine, he found that, ‘‘the anesthetic capability of cocaine is therefore a function of its acid group called by Paul Ehrlich the ‘anesthesiophoric’ group – the most potent being the benzoyl group’’.22 Today we know that the majority of clinically used local anaesthetics consist of a benzene ring linked via an amide or ester to an amine group, and their names still end in ‘caine’.
Differential and use‐dependent block by local anaesthetics
In those early days of anaesthesia a selective block of specific types of nerve fibre was noticed, termed ‘differential nerve block’, which was understood to be a sequential block of sensory information; a sequence of blockade of sharp pain, cold, warmth, touch, and finally conduction in motor fibres was considered most probable. A quantitative electrophysiological technique was employed by Gasser and Erlanger in 1929; they compared the different susceptibility of compound action potentials in nerves to pressure and cocaine‐containing solutions in accordance with their own classification based on fibre size and conduction velocity (from Aα‐ – fastest to C‐fibres – slowest).28 They suspected that diameter might be the main factor accounting for differential nerve block, based on the idea that the process responsible for impulse propagation was essentially similar in all fibres. With cocaine they observed that small fibres (slowly conducting) tended to be blocked before large ones, but in all cases a varying proportion of large fibres was blocked well before the compound action potential of small fibres had disappeared. That is why they concluded: ‘‘such a simple mechanism as has just been described should cause the fibres to be blocked systematically on a size basis; and since this does not rigidly hold the problem can be considered to be only partly solved. Some other as yet undetermined factor must be operating.’’28 Unfortunately these fundamental experiments with local anaesthetics did not report the block of C‐fibres, which are now known to contribute to dull pain. Later experiments with compound action potentials of peripheral nerves of various species revealed that C‐fibres are blocked partly when A‐fibres start to be blocked. However, the concentration necessary to half‐maximally block the C‐fibre compound action potential was 2–4 times higher than for A‐fibres, depending on the type of local anaesthetic used.295687 These data suggested that local anaesthetics with an ester structure have an inherently more potent action than those with an amide structure.
Experiments, especially in humans,476186 showed that C‐fibres themselves could be separated into different categories on the basis of their diverse physiological and electrical responses: alongside a large subgroup responding to mechanical, chemical and noxious stimuli which were termed polymodal receptors there was another subgroup that did not respond to these stimuli except after sensitization (e.g. with capsaicine, the main ingredient of hot pepper); these were called silent nociceptors.781 The existence of different categories of fibres raised another possible mechanism for differential local anaesthetic effects: different populations of ion channels. Recent findings support this idea and show that in small fibres, tetrodotoxin (TTX)‐resistant Na+ channels and Ca2+ channels are also present,3853 in addition to the classic Na+ channels sensitive to TTX. Their susceptibility to local anaesthetics will be described below, providing another possible explanation for the higher concentration of local anaesthetics needed to block all C‐fibres.
Block mechanisms
QX 314 is a quarternary derivative of the local anaesthetic lidocaine that has a permanent positive charge. It is not in clinical use but has interesting features that have helped to elucidate the mechanism of blocking Na+ channels. This drug blocks the channels only when applied to their intracellular side.24 Most local anaesthetics, except benzocaine, are amine compounds, which are charged at a pH below 6. The uncharged form is lipid soluble.34 Biophysical calculations based on the electrical field across the cell membrane88 suggested that the binding site is at a distance from the external surface of the membrane of approximately 60% of the membrane diameter (an estimate that agrees fascinatingly well with what we now know from the molecular structure; see section below). This finding gave rise to the first ideas about the blocking mechanism: the ‘receptor’ is in the pore; the charged form acts on the ‘receptor’, and the drug molecules have to pass through the lipid membrane to act. Another blocking characteristic was observed at that time: nearly full sized Na+ currents could be elicited during the first depolarizing impulse in the presence of a local anaesthetic, but subsequent impulses elicited smaller and smaller peak currents.74 It was suggested that the drug binds cumulatively and that this block needs open channels. This accumulation of inhibition has been called use‐dependent block or phasic block.2079 Subsequently, the guarded receptor hypothesis was proposed,72 suggesting that the receptor is protected in the pore and that the channel needs to be open before it can be blocked. The impact of use‐dependent block became manifest at higher firing frequencies of nerve fibres, where lower concentrations of local anaesthetics were needed to block compound action potentials (see Fig. 4).56
Structure of Na+ channels
Before we try to understand the mechanisms by which local anaesthetics block Na+ channels at the molecular level it is helpful to understand current thinking on the structure of the channels.
The Na+‐conducting part of the channel is called the α‐subunit,4546 a single polypeptide protein of relative molecular mass ∼260 000.1 The functional amino acid sequence encoded by the corresponding cDNA results in a ∼1950 amino acid chain which spans the cell membrane several times. The channel consists of four domains (DI–DIV; Fig. 1a),80 each containing six helices which span the membrane and are named the S1–S6 segments. The link between the S5 and S6 segments in each domain is of particular interest because these ‘pore loops’ form the outer pore and contain the amino acid sequence DEKA (in each domain). This structure represents the selectivity filter allowing primarily Na+ ions to pass through the pore (Fig. 1a). The region of the outer pore mouth is also involved in the binding of toxins such as TTX, batrachotoxin and conotoxins.687885 Interestingly, the binding site for batrachotoxin, although localized at the outer pore mouth, seems to influence the binding potency of local anaesthetics at the inner pore regions.168384 Two regions involved in gating were identified from early mutagenesis work with the Na+ channel. Firstly, as a voltage‐dependent channel, it needs a voltage sensor, which would have to be a charged region. The most obvious charged regions are the S4 transmembrane helices, which each contain a number of positively charged lysine and arginine residues. Substituting uncharged amino acids at these points reduces the voltage sensitivity, suggesting that the S4 helices are the voltage sensors.75
An intracellular link between DIII and DIV is important for Na+ channel function: it underlies ‘fast’ inactivation and will be described in the next section (Fig. 1b). Another type of inactivation, the slow ‘C‐type’,8 appears to involve the pore loops mentioned above. In addition to the pore‐forming α‐subunits, at least nine variants of which have been expressed (10 gene sequences are known17), there are three auxiliary subunits (β1–3). These β‐subunits have been shown to influence the activation and inactivation characteristics of expressed α‐subunits, and to affect the level of expression of channel proteins.4173743
Molecular functions of the Na+ channel
The simplest mechanistic scheme contains three states. First, there is the closed state at potentials below –70 mV. The pore in the channel is occluded so that Na+ ions cannot pass from one side to the other. Second, the open state of the channel is initiated by depolarization of the transmembrane potential to the threshold potential (usually above –40 mV). In response to depolarization, the channel opens within a millisecond and allows Na+ ions to diffuse down their concentration gradient through the pore, causing an inward current and depolarizing the transmembrane potential even further, which continues a self‐driven depolarization. This process underlies the upstroke of the action potential of most excitable cells. During channel opening the S4 segment twists back, driven by both the changed potential difference and intrinsic charge changes, which allow the outer pore mouth to expand, resulting in a 20° twist of the α‐helix.17 The third state follows activation during prolonged depolarization and is termed the inactivated state, because current inactivation is seen in macroscopic currents (Fig. 2). Thus, following depolarization the macroscopic Na+ current reaches a peak and then decreases as a function of time, often mono‐exponentially, even if the membrane potential is held at positive potentials. This self‐declining inward current, in addition to the outward current through voltage‐gated K+ channels, is one reason why repolarization occurs after a depolarization caused by an action potential. The inactivated state was shown to be a non‐conducting mode of the channel, although gating currents were measured.3 Application of enzymes such as pronase, applied from the internal side in internally perfused squid giant axons, prevented this ‘fast’ inactivation process. It was therefore concluded that the inactivation gate is positioned on the inner side of the channel protein.34 A sequence of three amino acids is important for ‘fast’ inactivation, and is called the IFM particle; it is the cytoplasmic linker between domains DIII and DIV (Fig. 1b).17 In ‘fast’ inactivation the IFM particle may function like a ‘lid’, plugging the pore (a ball on a chain is an alternative analogy) by binding to sites situated on or near the inner vestibule. The role of the IFM particle in binding of local anaesthetics is not fully understood but it seems that this ‘lid’ retains blockers inside the pore during use‐dependent blockade.517 Therefore Na+ ions can no longer pass through the pore, even though the pore is open at the outer mouth. The fast inactivated state is of interest to anaesthesia for two reasons: this state seems to play an important role in high‐affinity binding (discussed below), and the movement of the S4 segment in gating (important for activation and closing as described above) directly influences fast inactivation and vice versa.55
Molecular determinants of local anaesthetic action on Na+ channels
With the knowledge of the molecular structure of Na+ channels, especially the amino acid sequence of the pore‐forming α‐subunit, it became possible to uncover the ‘receptor’ for local anaesthetics. Experiments on the rat brain Na+ channel IIa (Nav 1.2) revealed that exchanging the amino acid phenylalanine (F) at position 1764 with alanine (A) made the channel (Fig. 1b) virtually insensitive to use‐dependent block. Measurements of the IC50 for the inactivated wild‐type Na+ channel with depolarizing prepulses revealed a 1:1 binding relationship, with 300‐fold smaller values compared with tonic blockade. In contrast, in the F1764A mutation the IC50 values for inactivated Na+ channels were only six times smaller than those for tonic blockade. Another mutant, Y1771A, exhibited less use‐dependent block and reduced drug binding at depolarized potentials, but the effect was smaller than for F1764A.55 These results led to a model for the ‘receptor’ site of local anaesthetics in the pore of the Na+ channel. The residues 1764 (F) and 1771 (Y) described above are hydrophobic aromatic residues separated by two turns on the same face of the protein helix of the pore‐forming S6 segment (Fig. 1b). These amino acids are about 11 Å apart, and most effective local anaesthetics are 10–15 Å in length. The local anaesthetics have positively charged moieties at either end, which could interact through hydrophobic or π electrons with these kinds of amino acid residues.933 The substitution with alanine changed the size and the chemical properties in these mutants, with minimal effect on the protein secondary structure57 and demonstrated that these are probably the main determinants of drug binding for the opened channel. This idea is supported by experiments with the permanently charged local anaesthetic QX314 which after intra‐oocyte injection displayed a use‐dependent block of more than 50% in expressed wild‐type Na+ channels but produced hardly any use‐dependent block in the F1764A mutant. In these types of experiment the only possible access route to this site was via the open channel; recovery times comparable with those in the wild‐type indicate that the escape pathway of the local anaesthetic is not altered.
Another mutation oriented towards the pore, I1760A (Fig. 1b), did not alter the local anaesthetic affinity of either the open or the inactivated state of the Na+ channel. Instead, the rate of drug dissociation from the mutant channel was found to be eight times faster than for wild‐type channels. This might be explained by the fact that the I1760A mutation is closer to the extracellular side on the S6 segment at the channel outer mouth, and isoleucine is a bulky residue. It therefore seems likely that the drugs could escape more easily from the mutated channel. Confirmation came from experiments with externally applied QX314, normally ineffective, which rapidly blocked this mutant. Thus, the mutation created a pathway for the extracellular drug to enter, supporting the idea that faster recovery from drug‐bound states with nearly unchanged use‐dependent block properties is the result of easier escape of the drug from the channel.55
An explanation emerges which identifies two binding sites in the pore of the brain Na+ channel (positions 1764 and 1771) whose hydrophobic parts interact with the corresponding regions of the local anaesthetic molecule. The residue oriented more towards the mouth of the pore (1760) guards the fast escape of the drug molecule to the extracellular side and protects the channel from extracellular drugs.
It should be noted that mutations in the S6 segment at positions other than those described above, I1761A, V1767A and N1769A, caused more pronounced blockade at holding potentials of –90 mV. The inactivation curve for these mutants was found to be shifted by 7–13 mV to more negative potentials, therefore the increased sensitivity might be caused by an increased proportion of inactivated Na+ channels. But even at more negative holding potentials where the inactivation was removed, it was found that mutations I1761A and V1766A were about three times more sensitive than wild‐type Na+ channels under resting conditions. The mutation N1769A at holding potentials of –140 mV (where all fast inactivation was removed completely) was 15 times more sensitive than wild‐type Na+ channels. The residues 1761 (I), 1766 (V) and 1769 (N) are oriented away from the inner pore towards the lipid phase of the membrane. It is therefore possible that mutations of these residues influence the increased sensitivity of the inner pore local anaesthetic receptor by an allosteric mechanism. It can also be imagined that these positions are responsible for the resting blockade, explaining the higher affinity of lipophilic drugs under resting conditions. In addition, one must consider the possibility that these mutations cause changes in the inactivation behaviour of the channels, perhaps by inducing the binding site in the pore in functionally inactivated binding confirmation.55
The same results have been obtained involving structurally similar locations in heart and muscle Na+ channels, explaining the absence of substantial differences in affinity of local anaesthetics for Na+ channel subtypes.5 Only the position numbers of the amino acids are different: for example 1579 and 1586 in rat skeletal muscle Na+ channels (Nav 1.4) correspond to positions 1764 and 1771, respectively, in rat brain Na+ channels (Nav 1.2).
DIII, the neighbour domain of D IV, also appears to be important for binding of local anaesthetics.8490 Point mutations in the S6 segment of these domains resulted in a reduced affinity of local anaesthetics for neuronal and muscle Na+ channels. This indicates that at least some drug molecules are bound primarily at the S6 segment in DIV and DI but that the molecule also binds on the other side of the channel pore at the S6 segment of DIII.
In one aspect benzocaine is different from the other local anaesthetic: it is the only clinically used local anaesthetic that is not charged (because of its low pKa). Yet it is still suggested that benzocaine shares the same binding site as other local anaesthetics,62 at least in the neuronal and muscle Na+ channels, and it could be speculated that it might be again in the inner pore. Admittedly, the low affinity of benzocaine compared with other local anaesthetics (IC50 ∼800 µM) and the missing use‐dependent block6282 do not suggest a very high affinity mechanism.
The small differences in potency and toxicity of enantiomers seems to have a counterpart on the molecular level. A stereoselectivity of around 1.5 was found for Na+ channels from skeletal muscle and human heart cells (Nav 1.5).44 Primarily in the human heart channel, it was demonstrated that the binding residues 1760 and 1765 (the corresponding positions in Nav 1.2 are 1764 and 1771) contribute to the weak stereoselectivity.
Tetrodotoxin‐resistant Na+ channels
The description of the molecular mechanism presented above has been derived mainly from Na+ channels that are sensitive to TTX in the nanomolar range, as well as the cardiac channel, which is less sensitive but not insensitive to TTX. Typical TTX‐resistant Na+ channels are found primarily in neurones of the dorsal root ganglia (DRG).2 They are termed SNS/PN3 and SNS2/NaN, in the new nomenclature Nav 1.8 and Nav 1.9, respectively.30596477 Apart from being some hundred times less sensitive to TTX, the current kinetics of these channels are slower than those of the ‘fast’ TTX‐sensitive Na+ currents and their thresholds of activation and half‐maximal inactivation are at more depolarized potentials (Fig. 2). Their distribution predominantly in small‐to‐medium sized DRG neurones connected to Aδ‐ and C‐fibres immediately suggested a functional role in pain conduction. Beside their presence in the soma, there is electrophysiological and immunohistochemical evidence for the presence of TTX‐resistant Na+ channels in peripheral nerve terminals233853 and in nociceptor nerve terminals.1373 Experiments1373 on sensory nerve fibres innervating the dural membrane clearly showed that 50% of small Aδ‐fibres and 85% of C‐fibres were unaffected by 1 µM TTX! This indicates that TTX‐resistant Na+ channels are important elements of signal transduction in the smallest nerve endings. Indeed, null mutant mice lacking the SNS/PN3 Na+ channel show partial analgesia to painful stimuli.21
The question had already been raised in previous studies whether this TTX‐resistant Na+ current is blocked by local anaesthetics. In fact, the blocking mechanisms were found to be very similar to those in TTX‐sensitive Na+ currents, but the main difference was a 2–6 times lower affinity of TTX‐resistant Na+ currents, depending on the type of local anaesthetic (Figs 3 and 4).105866 One study demonstrated that TTX‐resistant action potentials in small sensory neurones, which are involved in the generation and conduction of pain‐related impulses, are suppressed by lidocaine and bupivacaine in a concentration‐dependent manner at clinically relevant concentrations (Fig. 3).67 These concentrations were 3–4 times higher than those required to block TTX‐sensitive Na+ currents in the same type of neurone. Furthermore, sensitivity to local anaesthetics was higher by a factor of 10–20 in firing frequency than in amplitude of the TTX‐resistant action potentials (Fig. 4). This observation may be relevant for the analgesic properties of local anaesthetics, especially when considering that intensity of pain is encoded by the frequency of action potentials. In addition, it has been observed that the local anaesthetic concentration for half‐maximal blockade of ion currents does not predict the relevant concentration required for a functional blockade of neuronal signal conduction.
Potassium channels
As with voltage‐dependent Na+ channels, voltage‐dependent and voltage‐independent K+ channels3949 are blocked by local anaesthetics. This is of interest because in the peripheral nervous system K+ channels contribute to the falling phase of action potentials, determine the after‐spike period and play a part in setting the resting potential. Voltage‐gated K+ channels in particular have been investigated in dorsal horn neurones and DRG and partially in nerve fibres. The major difference between local anaesthetic action in voltage‐gated K+ currents compared with Na+ currents is the lower affinity in the former. Depending on the type of voltage‐dependent K+ current, the affinity for bupivacaine is 4‐ to 10‐fold lower and for lidocaine it is 10‐ to 80‐fold lower.1239 This inhibition of K+ channels might be an additional explanation for broadening of the action potential in the presence of local anaesthetics, because the voltage‐gated K+ currents contribute to the repolarization phase of action potentials.
A voltage‐independent K+ channel was also found to be blocked by local anaesthetics in sensory neurones.11 This ‘flicker’ K+ channel (so termed because of its flickering kinetics in high K+ solutions) is found mainly in thin myelinated fibres of frog sciatic nerves and was highly sensitive to local anaesthetics, requiring only 0.21 µM bupivacaine for half‐maximal blockade. This is the most sensitive ion channel in this preparation. Up to now this ‘flicker’ channel has not been detected in mammalian fibres.
In addition to these K+ channels, the ATP‐sensitive K+ channel in heart muscle cells has been found to be sensitive to lidocaine and bupivacaine.4850 This blockade of K+ channels may explain side‐effects and toxic effects of local anaesthetics in organs other than the peripheral nervous system, especially if the many subtypes of K+ channel have different sensitivities. Another mechanism was described for the Ca2+‐activated large conductance K+‐channel (BKCa), which was found to be activated by clinically relevant concentrations of ethanol (20–80 mM). The functional consequence of this activation in a subgroup of sensory neurones was shown to be a reduced action potential duration and a decrease of firing rate, which may partly explain the analgesic effect of ethanol.31
Calcium channels
Because the structure of voltage‐dependent Ca2+ channels closely resembles that of Na+ channels, it is not surprising that Ca2+ currents, especially in DRG, were found to be blocked by local anaesthetics.657689 A concentration– response curve for tetracaine in high‐voltage‐activated Ca2+ currents (L‐, N‐, Q‐ and P‐types) revealed an apparent dissociation constant of 80 µM. When specific blockers were used for L‐ and N‐type Ca2+ channels, it was found that the L‐type was more sensitive than either the N‐type Ca2+ channel or the remaining low‐voltage‐activated Ca2+ current (T‐type). Local anaesthetics other than tetracaine also depressed high‐voltage‐activated Ca2+ currents but were of different potency; the rank sequence was dibucaine > tetracaine > bupivacaine >> procaine = lidocaine. How could this finding be interpreted? Ca2+ channels are extremely important in synaptic transmission and in muscle cells, where they are involved in the coupling of electrical excitability with mechanical contraction. In human peripheral nerves, not only the central ends of the fibres in the dorsal horn but also some thin (mainly C) fibres contain Ca2+ channels.53 The functional consequences are suggested by a recent study in which tail‐flick latencies to radiant heat nociception were measured in the tails of mice to test the effect of subcutaneous infiltration of bupivacaine and Ca2+‐modulating drugs.70 It was found that an L‐type Ca2+ channel agonist reduced the duration of action and potency of bupivacaine anaesthesia. Nifedipine and nicardipine, antagonists of L‐type Ca2+ channels, increased the effects of bupivacaine. Other Ca2+ channel subtype blockers were also investigated but did not modify bupivacaine anaesthesia. Since there is no musculature in the tail, the results suggest that in some axons Ca2+ channels, at least in heat nociceptors in the skin, are sufficient to support propagation of action potentials.
Although the half‐maximal blockade of Ca2+ channels occurred at higher concentrations compared with Na+ channels (∼5–15 times), their blockade might contribute to interrupting the transmission at concentrations required for spinal anaesthesia. What this means for the synaptic transmission in the dorsal horn, which is triggered by Ca2+ channel opening, requires further investigation. This again raises the question of whether Ca2+ channels as targets of local anaesthetics underlie side‐effects or toxic effects in other organs. Besides these unwanted effects, there are some similarities between local anaesthetics and Ca2+ channel blockers (e.g. verapamil which showed a use dependence similar to local anaesthetics).71 This might be an interesting alternative target of the well‐known pro‐arrhythmic activity of some local anaesthetics on Na+ channels in heart muscle cells.
G‐proteins and G‐protein‐regulated channels
Although local anaesthetics are primarily considered to be blockers of ion channels, recent studies suggest a common intracellular site of action on different G‐protein‐coupled receptors.36 They showed that recombinant M1 muscarinic receptors expressed in oocytes were inhibited by local anaesthetics in a stereoselective and non‐competitive manner, suggesting a protein interaction. G‐protein α‐subunits involved in mediation of lysophosphatidic acid, M1, trypsin, and angiotensin AT1A receptor signalling were characterized. Lidocaine and its analogue QX314 were applied into oocytes expressing these receptors. Since the AT1A receptor, previously shown to be unaffected by local anaesthetics, was found not to signal via Gαq, but via Gαo and Gα14, the intracellular effect of local anaesthetics is most likely to be on the Gαq subunit. Blockade of the muscarinic M3 receptor was described by the same group, even though the M3 receptor lacks the extracellular terminus that explained the binding of local anaesthetics to M1 receptors.35
There is evidence that G‐protein‐gated inwardly rectifying K+ channels are blocked by local anaesthetics.69 In addition, it has been shown that bupivacaine mobilizes intracellular calcium ions via the inositol trisphosphate signalling cascade in sensory neurones.41 Taken together, these pathways might also explain why local anaesthetics exhibit side‐effects in other organs, and justify future work to elucidate whether they act in the same way in brain and heart.
General anaesthetics and ion channels
Clearly, the clinical effects of general and local anaesthetics are different. However, axonal ion conductance has been investigated as a possible mechanism of action of general anaesthetics.61532A partial blockade of mainly Na+ currents was found but this was not sufficient to suggest peripheral conduction blockade as a major mechanism. Compared with results with ligand‐gated ion channels in the central nervous system (described in more detail in other articles in this issue) the general anaesthetics are less potent at voltage‐gated ion channels. However, subsequent research has found that, depending on the species from which the channel originated, voltage‐gated currents can be affected at clinically relevant concentrations of potent inhalation anaesthetics.252764 For example, in Na+ channels the half‐maximal values of inactivation were shifted to more hyperpolarized values, resulting in fewer Na+ channels being available to be opened.
Besides voltage‐gated K+ channels, a class of leak or background K+‐selective channels is known. These channels are formed by two transmembrane segments with the conserved motif called the P domain and therefore named 2P domain K+ channels.51 Members of this class of 2P domain K+ channels, such as TREK‐1, TREK‐2, TASK‐1 and TASK‐2, have been reported to be activated by volatile anaesthetics. TASK‐1, in contrast to the other types, is also blocked by the local anaesthetic bupivacaine.14 The 2P domain K+ channels are not uniformly affected by volatile anaesthetics: TASK‐2 but not TASK‐1 is stimulated by chloroform, while TASK‐1 is partially inhibited by diethyl ether.52 Even though the stimulation of channel activity by halothane is specific, other 2P domain K+ channels—TWIK‐2, THIK‐1, TALK‐1 and TALK‐2—are inhibited.51 For channels sensitive to anaesthetics it was demonstrated that the C terminus but not the N terminus was crucial.52
Perspectives for local anaesthetics
Early hypotheses based on non‐specific interactions of lipid‐soluble anaesthetics with membrane bilayers have largely given way to the current idea that membrane‐associated proteins, particularly ion channels, are specifically modulated by local anaesthetics. Indeed, Na+ channels have been identified as a major target, with two different blocking mechanisms, tonic and phasic. The use‐dependent (phasic) block by local anaesthetics seems to be the mechanism that underlies the very high sensitivity of Na+ channels which is based on the binding of local anaesthetic molecules in the channel pore to few specific amino acids. A recent investigation tested a benzomorphan derivative, crobenetine (BIII 890 CL), which produces a very pronounced use‐dependence, with the phasic blockade being about 300 times more potent than the tonic blockade.16 Even though this agent was designed to protect the brain after permanent focal cerebral ischaemia, its highly use‐dependent Na+ channel block makes it a possible candidate as a local anaesthetic and for treatment of neuropathic pain.4042 Another specific suppression of pain might be expected from a drug that targets TTX‐resistant Na+ channels (e.g. Nav 1.8 and Nav 1.9), whose expression is confined to Aδ and C pain‐mediating fibres. However, the development of drugs that exhibit selective blockade of neuronal TTX‐resistant Na+ channels while leaving TTX‐sensitive channels unblocked is still to be accomplished.
Acknowledgements
I am grateful to Professor Werner Vogel and Dr Marco Gruß for critical comments on the manuscript, and Christoph Malik for his invaluable help with Figure 1.
Fig 1 Structure of a Na+ channel α‐subunit. (a) Schematic diagram of the four domains DI–DIV. Each domain consists of six segments, which span the membrane. Part of the ‘pore’ loops, the amino acid links between the S5 and the S6 segments are symbolized as red triangles. These four amino acid links ‘DEKA’ form the selectivity filter in the outer pore mouth (for more details see section Structure of Na+channel). (b) 3D sketches of a Na+ channel with top and bottom views and large cross‐section as derived from data of cryo‐electron microscopy and single particle analysis.60 On the left side of the large cross‐section the cut is through the S4 segment (the function of the cavity marked in red is unclear) and on the right side the cross‐section is through the S6 segment overlaid with the amino acid sequence of rat brain Na+ channel (Nav 1.2). Residues that are coloured and numbered (60 for amino acid 1760 etc.) are important for the affinity of local anaesthetics. (Adapted with permission from Nature18 and Mosby Saunders.63)
Fig 1 Structure of a Na+ channel α‐subunit. (a) Schematic diagram of the four domains DI–DIV. Each domain consists of six segments, which span the membrane. Part of the ‘pore’ loops, the amino acid links between the S5 and the S6 segments are symbolized as red triangles. These four amino acid links ‘DEKA’ form the selectivity filter in the outer pore mouth (for more details see section Structure of Na+channel). (b) 3D sketches of a Na+ channel with top and bottom views and large cross‐section as derived from data of cryo‐electron microscopy and single particle analysis.60 On the left side of the large cross‐section the cut is through the S4 segment (the function of the cavity marked in red is unclear) and on the right side the cross‐section is through the S6 segment overlaid with the amino acid sequence of rat brain Na+ channel (Nav 1.2). Residues that are coloured and numbered (60 for amino acid 1760 etc.) are important for the affinity of local anaesthetics. (Adapted with permission from Nature18 and Mosby Saunders.63)
Fig 2 Tetrodotoxin (TTX)‐resistant and TTX‐sensitive Na+ currents in rat dorsal root ganglion neurones blocked by lidocaine. (a) TTX‐resistant Na+ currents. Fitting to the decaying parts (dotted lines) showed time constants of 3.6 ms in Ringer‐TTX solution and 3.2 ms in the presence of lidocaine 300 µmol litre–1. (b) The TTX‐sensitive Na+ currents decayed faster, with time constants of 0.4 ms in Ringer solution and 0.5 ms in the presence of lidocaine 50 µmol litre–1. Less than 2% of the current remained in the presence of TTX 200 nmol litre–1. (Adapted with permission of J. Neurophysiol.66)
Fig 2 Tetrodotoxin (TTX)‐resistant and TTX‐sensitive Na+ currents in rat dorsal root ganglion neurones blocked by lidocaine. (a) TTX‐resistant Na+ currents. Fitting to the decaying parts (dotted lines) showed time constants of 3.6 ms in Ringer‐TTX solution and 3.2 ms in the presence of lidocaine 300 µmol litre–1. (b) The TTX‐sensitive Na+ currents decayed faster, with time constants of 0.4 ms in Ringer solution and 0.5 ms in the presence of lidocaine 50 µmol litre–1. Less than 2% of the current remained in the presence of TTX 200 nmol litre–1. (Adapted with permission of J. Neurophysiol.66)
Fig 3 Local anaesthetics reduce firing frequency in small dorsal root ganglia neurones. (a) Trains of tetrodotoxin (TTX)‐resistant action potentials elicited by a 750 ms 400 pA current stimulus are reduced in firing frequency by increasing concentrations of lidocaine. (b) Similar effects as in (a) seen with bupivacaine in another neurone. Extracellular solution in bath containing TTX 200 nM; high KI in pipette; 22–23°C. (Modified with permission from International Association of Pain.67)
Fig 3 Local anaesthetics reduce firing frequency in small dorsal root ganglia neurones. (a) Trains of tetrodotoxin (TTX)‐resistant action potentials elicited by a 750 ms 400 pA current stimulus are reduced in firing frequency by increasing concentrations of lidocaine. (b) Similar effects as in (a) seen with bupivacaine in another neurone. Extracellular solution in bath containing TTX 200 nM; high KI in pipette; 22–23°C. (Modified with permission from International Association of Pain.67)
Fig 4 Different sensitivities to local anaesthetics of firing frequency, action potential (AP) amplitude and tetrodotoxin (TTX)‐resistant Na+ current. (a) The number of TTX‐resistant (TTXr) AP (left ordinate) during 750 ms current injections depended on the concentration of lidocaine. A Hill equation was fitted to the data, revealing IC50 values of 24, 27 and 23 µM for 300, 400 and 500 pA current stimuli, respectively, from a single small dorsal root ganglion neurone. Blockade of active amplitudes of TTXr AP (right ordinate) revealed an IC50 of 730 µM lidocaine. For comparison, the reduction of TTXr Na+ current (dotted line66) is given, with an IC50 of 210 µM lidocaine. (b) The reduction in the number of AP from another neurone revealed IC50 values of 7, 9 and 7 µM bupivacaine for 200, 300 and 400 pA stimuli, respectively. The IC50 of reduction of active TTXr action potential amplitudes (open circles, same neurone) was 110 µM bupivacaine. The IC50 of reduction of TTXr Na+ current (dotted line66) was 32 µM bupivacaine. (Modified with permission from International Association of Pain.67)
Fig 4 Different sensitivities to local anaesthetics of firing frequency, action potential (AP) amplitude and tetrodotoxin (TTX)‐resistant Na+ current. (a) The number of TTX‐resistant (TTXr) AP (left ordinate) during 750 ms current injections depended on the concentration of lidocaine. A Hill equation was fitted to the data, revealing IC50 values of 24, 27 and 23 µM for 300, 400 and 500 pA current stimuli, respectively, from a single small dorsal root ganglion neurone. Blockade of active amplitudes of TTXr AP (right ordinate) revealed an IC50 of 730 µM lidocaine. For comparison, the reduction of TTXr Na+ current (dotted line66) is given, with an IC50 of 210 µM lidocaine. (b) The reduction in the number of AP from another neurone revealed IC50 values of 7, 9 and 7 µM bupivacaine for 200, 300 and 400 pA stimuli, respectively. The IC50 of reduction of active TTXr action potential amplitudes (open circles, same neurone) was 110 µM bupivacaine. The IC50 of reduction of TTXr Na+ current (dotted line66) was 32 µM bupivacaine. (Modified with permission from International Association of Pain.67)
References
Agnew WS, Levinson SR, Brabson JS, et al. Purification of the tetrodotoxin‐binding component associated with the voltage‐sensitive sodium channel from Electrophorus electricus electroplax membranes.1978
; : 2606
–10Akopian AN, Souslova V, England S, et al. The tetrodotoxin‐resistant sodium channel SNS has a specialized function in pain pathways. Neurosci
; 2
: –8Armstrong CM. Voltage‐dependent ion channels and their gating. 1992
; : S5
–13Baker MD, Wood JN. Involvement of Na+ channels in pain pathways. 2001
; : 27
–31Balser JR. The cardiac sodium channel: gating function and molecular pharmacology. 2001
; : 599
–613Bean BP, Shrager P, Goldstein DA. Modification of sodium and potassium channel gating kinetics by ether and halothane. 1981
; : 233
–53Belmonte C, Cervero F. Neurobiology of Nociceptors, 1st Edn. Oxford: Oxford University Press,
Benitah JP, Chen Z, Balser JR, et al. Molecular dynamics of the sodium channel pore vary with gating: interactions between P‐segment motions and inactivation. 1999
; : 1577
–85Bokesch PM, Post C, Strichartz G. Structure–activity relationship of lidocaine homologs producing tonic and frequency‐dependent impulse blockade in nerve. 1986
; : 773
–81Brau ME, Elliott JR. Local anaesthetic effects on tetrodotoxin‐resistant Na+ currents in rat dorsal root ganglion neurones. 1998
; : 80
–8Brau ME, Nau C, Hempelmann G, et al. Local anesthetics potently block a potential insensitive potassium channel in myelinated nerve. 1995
; : 485
–505Brau ME, Vogel W, Hempelmann G. Fundamental properties of local anesthetics: Half‐maximal blocking concentrations for tonic block of Na+ and K+ channels in peripheral nerve. 1998
; : 885
–9Brock JA, McLachlan EM, Belmonte C. Tetrodotoxin‐resistant impulses in single nociceptor nerve terminals in guinea‐pig cornea. 1998
; : 211
–7Buckler KJ, Williams BA, Honore E. An oxygen‐, acid‐ and anaesthetic‐sensitive TASK‐like background potassium channel in rat arterial chemoreceptor cells. 2000
; Pt 1: 135
–42Butterworth JF, Raymond SA, Roscoe RF. Effects of halothane and enflurane on firing threshold of frog myelinated axons. 1989
; : 493
–516Carter AJ, Grauert M, Pschorn U, et al. Potent blockade of sodium channels and protection of brain tissue from ischemia by BIII890CL. 2000
; : 4944
–9Catterall WA. From ionic currents to molecular mechanisms: the structure and function of voltage‐gated sodium channels. 2000
; : 13
–25Catterall WA. Structural biology – A 3D view of sodium channels. 2001
; : 988
–91Corning JL. Spinal anaesthesia and local medication of the cord. 1885
; : 183
–5Courtney KR. Mechanism of frequency‐dependent inhibition of sodium currents in frog myelinated nerve by the lidocaine derivative GEA. 1975
; : 225
–36Cummins TR, DibHajj SD, Black JA, et al. A novel persistent tetrodotoxin‐resistant sodium current in SNS‐null and wild‐type small primary sensory neurons. 1999
; : 1
–6Einhorn A. On the chemistry of local anesthetics. Munch
Wochenschr
; 46
: –20Fjell J, Hjelmstrom P, Hormuzdiar W, et al. Localization of the tetrodotoxin‐resistant sodium channel NaN in nociceptors. 2000
; : 199
–202Frazier DT, Narahashi T, Yamada M. The site of action and active form of local anesthetics. II. Experiments with quaternary compounds. 1970
; : 45
–51Frenkel C, Wartenberg HC, Rehberg B, et al. Interactions of ethanol with single human brain sodium channels. Res
1997
; : 113
–20Freud S. Über Coca. Ges
1884
; : 289
–314Friederich P, Benzenberg D, Trellakis S, et al. Interaction of volatile anesthetics with human Kv channels in relation to clinical concentrations. 2001
; : 954
–8Gasser HS, Erlanger J. Role of fibre size in the establishment of nerve block by pressure or cocaine. 1929
; : 581
–91Gissen AJ, Covino BG, Gregus J. Differential sensitivities of mammalian nerve fibers to local anesthetic agents. 1980
; : 467
–74Goldin AL, Barchi RL, Caldwell JH, et al. Nomenclature of voltage‐gated sodium channels. 2000
; : 365
–8Gruss M, Henrich M, König P, et al. Ethanol reduces excitability in a subgroup of primary sensory neurons by activation of BKCa channels. 2001
; : 1246
–56Haydon DA, Urban BW. The effects of some inhalation anaesthetics on the sodium current of the squid giant axon. 1983
; : 429
–39Heginbotham L, Abramson T, MacKinnon R. A functional connection between the pores of distantly related ion channels as revealed by mutant K+ channels. 1992
; : 1152
–5Hille B. Ionic Channels of Excitable Membranes, 3rd Edn. Sunderland, Massachusetts: Sinauer Associates, Inc.,
Hollmann MW, Ritter CH, Henle P, et al. Inhibition of m3 muscarinic acetylcholine receptors by local anaesthetics. 2001
; : 207
–16Hollmann MW, Wieczorek KS, Berger A, et al. Local anesthetic inhibition of G protein‐coupled receptor signaling by interference with Galpha(q) protein function. 2001
; : 294
–301Isom LL, De Jongh KS, Patton DE, et al. Primary structure and functional expression of the beta 1 subunit of the rat brain sodium channel. 1992
; : 839
–42Kobayashi JI, Ohta M, Terada Y. Tetrodotoxin resistant Na+ spikes of c fibers have at least two subtypes in the isolated bullfrog sciatic nerve. 1996
; : 9
–12Komai H, McDowell TS. Local anesthetic inhibition of voltage‐activated potassium currents in rat dorsal root ganglion neurons. 2001
; : 1089
–95Krause U, Weiser T, Carter AJ, et al. Potent, use‐dependent blockade of TTX‐resistant Na+‐channels in dorsal root ganglion neurones by BIII 890 CL. J
2000
; : S387
Kuboyama N, Nakoa S, Moriya Y, et al. Bupivacaine‐included Ca2+ release on intracellular Ca2+ stores in rat DRG neurones. 1997
; : 1
–103Laird JMA, Carter AJ, Grauert M, Cervero F. Analgesic activity of a novel use‐dependent sodium channel blocker, crobenetine, in mono‐arthritic rats. 2002
; in pressMalhotra JD, Kazen‐Gillespie K, Hortsch M, et al. Sodium channel beta subunits mediate homophilic cell adhesion and recruit ankyrin to points of cell‐cell contact. 2000
; : 11383
–8Nau C, Wang SY, Strichartz GR, et al. Block of human heart hH1 sodium channels by the enantiomers of bupivacaine. 2000
; : 1022
–33Noda M. Structure and function of sodium channels. 1993
; : 20
–37Noda M, Ikeda T, Suzuki H, et al. Expression of functional sodium channels from cloned cDNA. 1986
; : 826
–8Ochoa JL,Torebjork HE. Paraesthesiae from ectopic impulse generation in human sensory nerves. 1980
; : 835
–53Olschewski A, Brau ME, Olschewski H, et al. ATP‐dependent potassium channel in rat cardiomyocytes is blocked by lidocaine. Possible impact on the antiarrhythmic action of lidocaine. 1996
; : 656
–9Olschewski A, Hempelmann G, Vogel W, et al. Blockade of Na+ and K+ currents by local anesthetics in the dorsal horn neurons of the spinal cord. 1998
; : 172
–9Olschewski A, Olschewski H, Brau ME, et al. Effect of bupivacaine on ATP‐dependent potassium channels in rat cardiomyocytes. 1999
; : 435
–8Patel AJ, Honore E. Properties and modulation of mammalian 2P domain K+ channels. Neurosci
; 24
: –46Patel AJ, Honore E, Lesage F, et al. Inhalational anesthetics activate two‐pore‐domain background K+ channels. Neurosci
; 2
: –6Quasthoff S, Grosskreutz J, Schroder JM, et al. Calcium potentials and tetrodotoxin‐resistant sodium potentials in unmyelinated C fibres of biopsied human sural nerve. 1995
; : 955
–65Quincke H. Die Lumbalpunktion des Hydrozephalus. Klin Wochensch
r; 28
: –33Ragsdale DS, McPhee JC, Scheuer T, et al. Molecular determinants of state‐dependent block of Na+ channels by local anesthetics. 1994
; : 1724
–8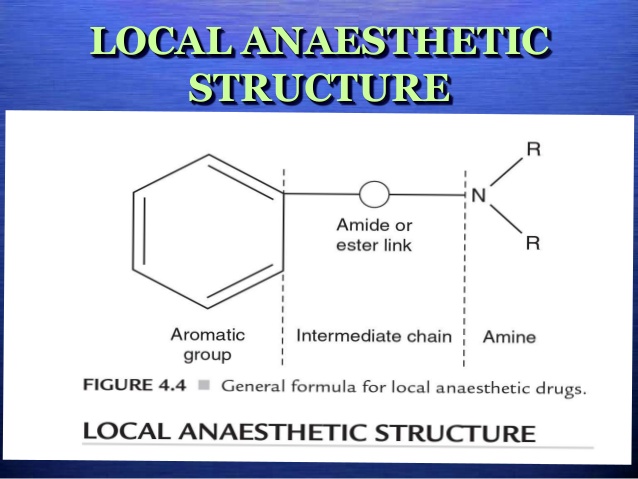
95
–165Richardson JS. The anatomy and taxonomy of protein structure. 1981
; : 167
–339Roy ML, Narahashi T. Differential properties of tetrodotoxin‐sensitive and tetrodotoxin‐resistant sodium channels in rat dorsal root ganglion neurons. 1992
; : 2104
–11Rush AM, Brau ME, Elliott AA, et al. Electrophysiological properties of sodium current subtypes in small cells from adult rat dorsal root ganglia. 1998
; : 771
–89Sato C, Ueno Y, Asai K, et al. The voltage‐sensitive sodium channel is a bell‐shaped molecule with several cavities. 2001
; : 1047
–51Schmelz M, Schmidt R, Bickel A, et al. Specific C‐receptors for itch in human skin. 1997
; : 8003
–8Schmidtmayer J, Ulbricht W. Interaction of lidocaine and benzocaine in blocking sodium channels. 1980
; : 47
–54Scholz A. Ion channel ligands/sodium channel ligands/local anesthetics: Mechanism of action. In: Evers AS, Maze M eds. Anesthetic Pharmacology and Physiology: Principles and Practice. Harcourt, Orlando: Mosby & Saunders, , in press
Scholz A, Appel N, Vogel W. Two types of TTX‐resistant and one TTX‐sensitive Na+ channel in rat dorsal root ganglion neurons and their blockade by halothane. J
1998
; : 2547
–56Scholz A, Kuboyama N, Bischoff U, et al. Local anesthetics and ketamine are inhibiting Ca2+ channels in rat DRG neurones. Arch
; 426
: Scholz A, Kuboyama N, Hempelmann G, et al. Complex blockade of TTX‐resistant Na+ currents by lidocaine and bupivacaine reduce firing frequency in DRG neurons. Neurophysiol
; 79
: –54Scholz A, Vogel W. Tetrodotoxin‐resistant action potentials in dorsal root ganglion neurons are blocked by local anesthetics. 2000
; : 47
–52Shon KJ, Olivera BM, Watkins M, et al. mu‐Conotoxin PIIIA, a new peptide for discriminating among tetrodotoxin‐sensitive Na channel subtypes. Neurosci
; 18
: –81Slesinger PA. Ion selectivity filter regulates local anesthetic inhibition of G‐protein‐gated inwardly rectifying K+ channels. J
; 80
: –18Smith FL, Davis RW, Carter R. Influence of voltage‐sensitive Ca++ channel drugs on bupivacaine infiltration anesthesia in mice. 2001
; : 1189
–97Sperelakis N. Electrophysiology of calcium antagonists. 1987
; Suppl 2: 19
–47Starmer CF, Grant AO, Strauss HC. Mechanisms of use‐dependent block of sodium channels in excitable membranes by local anesthetics. J
; 46
: –27Strassman AM, Raymond SA. Electrophysiological evidence for tetrodotoxin‐resistant sodium channels in slowly conducting dural sensory fibers. 1999
; : 413
–24Strichartz GR. The inhibition of sodium currents in myelinated nerve by quaternary derivatives of lidocaine. 1973
; : 37
–57Local Anesthesia Injection Names
Stuhmer W, Conti F, Suzuki H, et al. Structural parts involved in activation and inactivation of the sodium channel.1989
; : 597
–603Sugiyama K, Muteki T. Local anesthetics depress the calcium current of rat sensory neurons in culture. 1994
; : 1369
–78Tate S, Benn S, Hick C, et al. Two sodium channels contribute to the TTX‐R sodium current in primary sensory neurons. Neurosci
; 1
: –5Terlau H, Heinemann SH, Stuhmer W, et al. Mapping the site of block by tetrodotoxin and saxitoxin of sodium channel II. Lett
; 293
: –6Ulbricht W. Kinetics of drug action and equilibrium results at the node of Ranvier. 1981
; : 785
–828Vassilev PM, Scheuer T, Catterall WA. Identification of an intracellular peptide segment involved in sodium channel inactivation. 1988
; : 1658
–61Wall PD, Melzack R. Textbook of Pain, 4th Edn. Edinburgh, London, Melbourne, New York: Churchill Livingstone, 1999
Wang GK, Quan C, Wang S. A common local anesthetic receptor for benzocaine and etidocaine in voltage‐gated mu1 Na+ channels. Arch
; 435
: –302Wang SY, Barile M, Wang GK. Disparate role of Na+ channel D2‐S6 residues in batrachotoxin and local anesthetic action. 2001
; : 1100
–7Wang SY, Nau C, Wang GK. Residues in Na+ channel D3–S6 segment modulate both batrachotoxin and local anesthetic affinities. J
; 79
: –87Wang SY, Wang GK. Batrachotoxin‐resistant Na+ channels derived from point mutations in transmembrane segment D4–S6. J
; 76
: –9Weidner C, Schmelz M, Schmidt R, et al. Functional attributes discriminating mechano‐insensitive and mechano‐responsive C nociceptors in human skin. Neurosci
; 19
: –90Wildsmith JA, Gissen AJ, Takman B, et al. Differential nerve blockade: esters v. amides and the influence of pKa. 1987
; : 379
–84Woodhull AM. Ionic blockage of sodium channels in nerve. 1973
; : 687
–708Xiong ZL, Strichartz GR. Inhibition by local anesthetics of Ca2+ channels in rat anterior pituitary cells. 1998
; : 81
–90Yarov‐Yarovoy V, Brown J, Sharp EM, et al. Molecular determinants of voltage‐dependent gating and binding of pore‐ blocking drugs in transmembrane segment IIIS6 of the Na(+) channel alpha subunit. 2001
; : 20
–7doi: 10.2344/0003-3006-59.2.90
PMID: 22822998
This article has been cited by other articles in PMC.
Abstract
Local anesthetics have an impressive history of efficacy and safety in medical and dental practice. Their use is so routine, and adverse effects are so infrequent, that providers may understandably overlook many of their pharmacotherapeutic principles. The purpose of this continuing education article is to provide a review and update of essential pharmacology for the various local anesthetic formulations in current use. Technical considerations will be addressed in a subsequent article.
Keywords: Local anesthetics, Pharmacology, Drug toxicity, Dentistry
Local anesthetics interrupt neural conduction by inhibiting the influx of sodium ions through channels or ionophores within neuronal membranes. Normally these channels exist in a resting state, during which sodium ions are denied entry. When the neuron is stimulated, the channel assumes an activated or open state, in which sodium ions diffuse into the cell, initiating depolarization. Following this sudden change in membrane voltage, the sodium channel assumes an inactivated state, during which further influx is denied while active transport mechanisms return sodium ions to the exterior. Following this repolarization, the channel assumes its normal resting state. An appreciation of these sodium channel states helps to explain the preferential sensitivity of local anesthetics for various classes of neuronal fibers.
Local anesthetics have greater affinity for receptors within sodium channels during their activated and inactivated states than when they are in their resting states.1,2 Therefore, neural fibers having more rapid firing rates are most susceptible to local anesthetic action. Also, smaller fibers are generally more susceptible, because a given volume of local anesthetic solution can more readily block the requisite number of sodium channels for impulse transmission to be entirely interrupted. For these reasons the tiny, rapid-firing autonomic fibers are most sensitive, followed by sensory fibers and finally somatic motor fibers.1,2 The anesthesiologist blocking mixed spinal nerves is acutely aware of these differential sensitivities. As patients recover from spinal anesthesia they first regain voluntary motor function, then sensation returns, and finally they can micturate (autonomic control). The dentist is generally spared this consideration because the trigeminal nerve branches anesthetized for dental procedures are comprised only of small, rapid-firing sensory fibers. However, the many classes of sensory fibers also vary in their diameters and firing rates. For example, pain fibers are more sensitive than those carrying pressure and proprioception. A patient may remain disturbed by a sense of pressure despite complete anesthesia of pain fibers.
GENERAL PROPERTIES OF LOCAL ANESTHETICS
The molecular structure of all local anesthetics consists of 3 components: (a) lipophilic aromatic ring, (b) intermediate ester or amide linkage, and (c) tertiary amine. Each of these components contributes distinct clinical properties to the molecule. (See Figure 1.)
Local anesthetic structure.
Anesthetic Potency
Local anesthetics vary in their potency, allowing for concentrations that range typically from 0.5 to 4%. This is largely the result of differences in lipid solubility, which enhances diffusion through nerve sheaths and neural membranes. This property is determined by the aromatic ring and its substitutions, along with those added to the tertiary amine. For example, bupivacaine is more lipid soluble and potent than articaine, allowing it to be formulated as a 0.5% concentration (5 mg/mL) rather than a 4% concentration (40 mg/mL).
Time for Onset
Greater lipid solubility of a drug not only enhances potency but also enables more rapid diffusion through cell membranes. For local anesthetics, this hastens the onset for anesthesia in isolated fibers during in vitro studies, but it must be appreciated that other factors come into play clinically. For example, inherent vasodilating properties may promote systemic absorption before the anesthetic reaches the nerve membrane. High lipid solubility may impede dispersion throughout tissue fluids and also fosters sequestration in neighboring adipose tissues or myelin sheaths. In either case, fewer numbers of molecules reach the neuronal membrane and onset is delayed. Therefore, unlike in vitro studies of isolated fibers, greater lipid solubility generally slows the onset of anesthesia in the clinical setting. Injecting higher concentrations that allow a greater number of molecules to reach the membrane and hasten onset can offset this influence. Although bupivacaine and articaine are both highly lipid soluble, the 4% concentration of articaine provides for a much faster onset.
Despite myriad factors that influence the quantity of local anesthetic reaching the nerve fibers, the most important factor that determines the onset of anesthesia is the proportion of these molecules that exist in a lipid-soluble rather than a water-soluble state. The terminal amine illustrated in Figure 1 may exist in a tertiary form (3 bonds) that is lipid soluble, or as a quaternary form (4 bonds) that is positively charged and renders the molecule water soluble. For the local anesthetic base to be stable in solution, it is formulated as a hydrochloride salt. As such, the molecules exist in a quaternary, water-soluble state at the time of injection and are unable to penetrate the neuron. Therefore the time for onset of local anesthesia is directly related to the proportion of molecules that convert to the tertiary, lipid-soluble structure when exposed to physiologic pH (7.4). This proportion is determined by the ionization constant (pKa) for the anesthetic and is calculated using the Henderson-Hasselbalch equation:
In simpler terms, if a local anesthetic were to have a pKa of 7.4 and to be injected into tissues having a physiologic pH of 7.4, 50% of the molecules would exist in the quaternary (cationic) form and 50% would exist in the tertiary (uncharged) form; only half the molecules would be lipid soluble and able to penetrate the neuron. Unfortunately, the pKa for all local anesthetics is greater than 7.4 (physiologic pH), and therefore a greater proportion of the molecules exist in the quaternary, water-soluble form when injected into normal tissue. The clinical caveat is that the higher the pKa for a local anesthetic, the fewer molecules are available in their lipid-soluble form. This will delay onset. Furthermore, the acidic environment associated with inflamed tissues lowers their pH well below 7.4 and favors the quaternary, water-soluble configuration even further. This has been suggested as one explanation for difficulty when attempting to anesthetize inflamed or infected tissues.1,2 In these situations, for example, bupivacaine (pKa 8.1) would be less desirable than mepivacaine (pKa 7.6).
It must be clarified, however, that once the tertiary molecules enter the neuron, they reionize to the quaternary form, which is credited with the actual blockade of the sodium channel. The sequence of events that leads to neural blockade is illustrated in Figure 2.
Local anesthetic action. An injected local anesthetic exists in equilibrium as a quaternary salt (BH+) and tertiary base (B). The proportion of each is determined by the pKa of the anesthetic and the pH of the tissue. The lipid-soluble base (B) is essential for penetration of both the epineurium and neuronal membrane. Once the molecule reaches the axoplasm of the neuron, the amine gains a hydrogen ion, and this ionized, quaternary form (BH+) is responsible for the actual blockade of the sodium channel. The equilibrium between (BH+) and (B) is determined by the pH of the tissues and the pKa of the anesthetic (pH/pKa).
Metabolism and Elimination
The intermediate chain or linkage provides a convenient basis for classification of local anesthetics, and also determines their pattern of elimination. Amides are biotransformed in the liver but esters are hydrolyzed in the bloodstream by plasma esterases. Ester local anesthetics are no longer packaged in dental cartridges and are used infrequently, with the exception of benzocaine, found in several topical anesthetic preparations. Articaine is unique in this regard. It is classified as an amide according to its intermediate linkage, but also contains an ester side chain on its aromatic ring. Hydrolysis of this side chain renders the molecule inactive, and it is therefore eliminated in a manner identical to ester anesthetics.
Duration of Action
Local anesthetics vary in their duration of action due primarily to differences in their affinity for protein. Like most drugs, local anesthetics reversibly bind to plasma proteins while circulating in the bloodstream. This property is expressed as the percentage of circulating drug that is protein bound and has been found to correlate with an anesthetic's affinity for protein within sodium channels as well. The greater the tendency for protein binding, the longer the anesthetic will sustain neural blockade. For example, bupivacaine exhibits 95% protein binding compared to 55% for mepivacaine, and this is credited for the difference in their duration of neural blockade.
Duration of anesthesia is also influenced by the time a local anesthetic remains in close proximity to neural fibers. Sequestration of highly lipid-soluble anesthetics locally may allow for continual release to the neuronal membranes, prolonging duration, but constriction of neighboring vasculature is more significant in this regard. For this reason, vasopressors are added to many formulations in order to delay absorption and prolong anesthesia. This is particularly important because local anesthetics themselves vary in their ability to produce vasodilation. For example, when used without vasopressors, lidocaine shortens its own duration by dilating local vasculature, whereas mepivacaine and bupivacaine do not. Plain lidocaine formulations may be useful for brief procedures following infiltration, but their efficacy for nerve block is poor.
LOCAL ANESTHETIC TOXICITY
Systemic toxicity attributed to local anesthetics is dose dependent, but an understanding of these doses is not always a simple matter. The use of anesthetic cartridges in dentistry has unfortunately spawned carelessness in appreciating the actual amount of anesthetic we administer to our patients. Regrettably, this practice continues to be nurtured during undergraduate training and in many well-respected dental publications. A dental cartridge represents a volume, not a dose that is more properly expressed as milligrams or micrograms. Moreover, dental cartridges often contain 2 drugs: a local anesthetic and a vasopressor, each having a separate dose. Further complicating matters, dental cartridges contain peculiar volumes such as 1.7 or 1.8 mL. The sum of these issues makes actual dosage calculations trying and lends itself to memorization of amounts per cartridge rather than actual appreciation of proper doses. This practice becomes further complicated when cartridges contain various concentrations of local anesthetics and vasopressors. To simplify dosage calculations, it is wise to abort the concept of cartridges and consider each to contain 2 mL of volume. This will overestimate the amount administered to a patient, which is a safe practice. For example, when 4½ cartridges have been administered, estimate it as 9 mL. This unit of volume can be more easily converted to the approximate dose of each drug in milligrams or micrograms as illustrated in Table 1.
Table 1.
As local anesthetics are absorbed from the injection site, their concentration in the bloodstream rises and the peripheral nervous system and central nervous system (CNS) are depressed in a dose-dependent manner. (See Figure 3.) Low serum concentrations are used clinically for suppressing cardiac arrhythmias and status seizures, but ironically, higher concentrations induce seizure activity. Convulsive seizures are the initial life-threatening consequence of local anesthetic overdose. Presumably this is due to selective depression of central inhibitory tracts, which allow excitatory tracts to run amuck. As serum concentrations continue to rise further, all pathways are inhibited, resulting in coma, respiratory arrest, and eventually cardiovascular collapse. Evidence of lidocaine toxicity may commence at concentrations >5 µg/mL, but convulsive seizures generally require concentrations >10 µg/mL.
Approximate serum concentrations and systemic influences of lidocaine.
It is essential that local anesthetics be respected as CNS depressants, and they potentiate any respiratory depression associated with sedatives and opioids. Furthermore, serum concentrations required to produce seizures are lower if hypercarbia (elevated carbon dioxide) is present. This is the case when respiratory depression is produced by concurrent administration of sedatives and opioids. Goodson and Moore have documented catastrophic consequences of this drug interaction in pediatric patients receiving procedural sedation, along with excessive dosages of local anesthetics.
Although all local anesthetics carry comparable risk for CNS toxicity, it should be noted that bupivacaine exhibits greater potential for direct cardiac toxicity than other agents.1,2 The explanation is not fully established, but is thought to be related to the fact that bupivacaine has greater affinity for the inactive and resting sodium channel configurations and dissociates from these channels more slowly. This delays recovery from action potentials, rendering cardiac tissues susceptible to arrhythmias. This concern is relevant for certain medical procedures, during which bupivacaine is administered in very high doses. It has never been found to occur with doses up to the maximum recommended in dental anesthesia.
The obvious question is what systemic serum concentration follows administration of a particular dose of local anesthetic. In 1972, Scott et al published one in a series of landmark clinical studies assessing variables that determine subsequent concentrations of lidocaine and prilocaine in serum. It is not surprising that serum concentrations were found to vary according to the relative vascularity of the tissues in which the anesthetic was injected. Using lidocaine 400 mg, the highest serum levels illustrated in Figure 4 followed infiltration of vaginal mucosa and the lowest followed subcutaneous abdominal infiltration. In each case, however, peak serum level occurred 20–30 minutes following injection of lidocaine alone. Regardless of the route of administration, peak levels were reduced and the rate of absorption was delayed by adding epinephrine 1 ∶ 200,000 to the local anesthetic solution. It is reasonable to assume that systemic concentrations following submucosal injection in the oral cavity would approximate those following injection into vaginal mucosa because of similar vascularity. Unfortunately, there are very few dental studies that address higher doses of local anesthetics. However, Hersh et al have published an impressive study that found comparable results following multiple intraoral injections totaling 7 cartridges (1.7 mL each = ∼480 mg) of articaine containing epinephrine 1 ∶ 200,000. (See Figure 4.) One can reasonably conclude that adhering to published maximum recommended dosages for local anesthetics will not result in systemic serum levels that approach those associated with toxicity.
Local anesthetic serum concentrations. (See text for explanation. Adapted from Scott et al and Hersh et al.)
Additional variables were also addressed by Scott et al. As expected, the dosage and speed of injection were directly related to serum concentration. A solution's concentration, eg, 2 versus 4%, was not relevant; serum concentrations were related to the total dosage. Administering 20 mL of 2% or 10 mL of 4% (400 mg) produced the same serum concentration. When using lidocaine or other anesthetics, regardless of their formulated concentration, one must consider the dosage (milligrams) administered, not the volume (milliliters or cartridges).
Contrary to conventional thought, the age or weight of a patient does not predict systemic serum concentration following doses calculated as milligrams per age (years) or milligrams per kilogram. However, when managing pediatric patients, maximum dosages are conventionally expressed in mg/kg, and this should be followed as a precaution. It is of little relevance for adults, however, and one should adhere to guidelines expressed as maximum dose in milligrams, regardless of weight or age. Obviously, this maximum amount should not be exceeded when calculating mg/kg doses for large children.
When considering the toxicity of any drug class, one should be mindful of metabolites, as well as the parent drug. A metabolite of prilocaine, o-toluidine, can oxidize the iron in hemoglobin from ferrous (Fe2+) to ferric (Fe3+). Hemes so altered do not bind oxygen and normal hemes on the same hemoglobin molecule do not readily release their oxygen. This form of hemoglobin is called methemoglobin, and when >1% of total hemoglobin is so altered, the condition is called methemoglobinemia. Patients appear cyanotic and become symptomatic when the proportion of methemoglobin exceeds 15%.6 Hemoglobin saturation by pulse oximetry (SpO2) will decline despite clinical evidence of effective oxygenation and ventilation. For example, pulse oximeter readings may be <90%, but actual arterial oxygen tension (PaO2) may be within normal range (>80 mm Hg). The condition becomes life threatening when methemoglobin levels exceed 50–60%, and it is managed using intravenous methylene blue, which reduces the hemes to their normal state. Methemoglobinemia attributed to prilocaine is unlikely to follow the administration of recommended doses. Rarely, one may encounter a patient with hereditary methemoglobinemia, which contraindicates the use of prilocaine.
Allergy to Local Anesthetics
It is not unusual for patients to claim they are allergic to local anesthetics. Upon careful questioning, however, one generally finds that what they experienced was either a syncopal episode associated with the injection, or cardiac palpitations attributed to epinephrine contained either in the solution or released endogenously. Allergic reactions following local anesthetic injections are more likely attributable to preservatives (methylparaben) or antioxidants (sulfites) contained in the solution.7 Methylparaben is included in multidose vials to prevent microbial growth. It is no longer found in single-dose vials or dental cartridges. Sulfites prevent the oxidation of vasopressors and are included only in those dental cartridges containing epinephrine or levonordefrin.
Allergic reactions are triggered by immune mechanisms whereby lymphocytes are sensitized to antigen and, upon subsequent exposure, mediate a series of pathophysiologic changes. Gell and Coombs first categorized hypersensitivity (allergic) reactions as Type I through IV, based on distinct immunologic mechanisms.8 Type I reactions occur within minutes of provocation and are mediated by antibodies or immunoglobulin E (IgE) produced by B lymphocytes. This is the type most commonly provoked by components of local anesthetic formulations. Type 4 reactions are delayed for several days following provocation and are mediated by sensitized T lymphocytes. This type of reaction to local anesthetics has been implicated only rarely.
For drugs to be immunogenic, they must be of large molecular weight and possess multiple valences to be recognized by the immune cells.9 Large proteins such as animal-derived insulin fulfill these requirements and are well established as immunogenic. Most drug molecules are too small and actually combine with other molecules that act as carriers to induce an allergic reaction. In the case of sulfonamide antibiotics, for example, the phenyl ring containing an amine substitution is the perpetrator in the formation of the immunogenic complex. This moiety is common to other derivatives of para-aminobenzoic acid (PABA) such as methylparaben and some, but not all, ester local anesthetics. In these cases there may be the potential for cross-allergenicity because they have this molecular structure in common, eg, sulfa antibiotics, methylparaben, and esters of PABA.
It is careless to describe esters as more allergenic than amides when discussing local anesthetics. An ester is merely a chemical linkage and imparts no immunogenicity to a compound. Rather, it is a molecular component joined by this linkage that is the culprit. This misconception has caused several agents to be inaccurately labeled as cross-allergenic with sulfonamide antibiotics. Articaine is classified as an amide local anesthetic because of the linkage between its lipid-soluble ring and terminal amine. Its thiophene ring contains a sulfur atom, which has no immunogenic property, and an ester side chain that renders the compound inactive following hydrolysis. However, articaine does not liberate a metabolite resembling PABA and does not introduce concern regarding cross-immunogenicity with sulfonamides. In contrast, procaine is representative of esters derived from PABA and hydrolysis liberates a moiety that is potentially immunogenic (Figure 5).
Molecular structures and allergenicity. Immunogenicity is attributable to medications having a phenyl ring with a para-amine substitution. This is found in sulfonamide antibiotics and compounds containing para-aminobenzoic acid (PABA) such as certain sunscreens and cosmetics. It is also found in methylparaben preservatives and ester local anesthetics such as procaine. Ester linkages (procaine) or side chains (articaine) are not immunogenic, nor is the sulfur atom of a thiophene ring (articaine). * indicates immunogenic moiety.
A final misconception pertains to sulfites. These are included in local anesthetic solutions containing vasopressors to prevent their oxidation. They are inorganic compounds (−SO3) that have been implicated in allergic reactions, but they have no relation to immunogenicity attributed to PABA-related compounds. These agents are also used as antioxidants in fresh fruits and vegetables to preserve their color and overall appearance. It is significant that patients claiming allergy to such foods may experience cross-reactions with local anesthetic solutions containing vasopressors because they contain these same sulfites.
Reports of allergic reactions to local anesthetics have appeared in the scientific literature with some frequency., However, it is difficult to comprehend the accuracy or actual frequency because of inconsistency in methods of confirmation that include skin prick testing, intradermal injections, and drug provocative challenges. In many cases there has been no confirmation of the actual culprit, preservative or actual local anesthetic. Furthermore, only a very few have actually confirmed the presence of IgE to the offending drug by immunoblot testing. An extensive analysis of this literature has recently been provided by Speca et al.
In virtually all case reports, patients did indeed experience signs and symptoms consistent with an allergic reaction. Whether the actual pathogenesis was truly immune mediated (allergy) is probably more academic than pragmatic. The final event in these reactions is attributed to the synthesis and release of mediators referred to collectively as autacoids, of which histamine and leukotrienes are most significant. These autacoids not only produce direct effects on tissues but may also recruit various inflammatory cells that contribute to a so-called late-phase response that may not appear for days following provocation. Indeed, it is not uncommon for drugs to generate these autacoids by actions that are not immune mediated and therefore are not correctly classified as allergy. Meperidine stimulates release of histamine from mast cells, and nonsteroidal anti-inflammatory drugs may promote synthesis of leukotrienes. In such cases the patient's response has been conventionally labeled as pseudoallergic, to distinguish it from true allergy, which is immune mediated.
If a patient describes a reaction that is at least clinically consistent with allergy, the dentist should avoid using the offending agent until evaluated by an allergist. In the event an anesthetic is required before medical clearance can be obtained, the wisest choice would be either mepivacaine or prilocaine without vasopressors. Conventional wisdom holds that, if local anesthetics do indeed produce allergies, esters of PABA would be more likely than amide local anesthetics. Furthermore, by avoiding those solutions containing vasopressors, one avoids any bisulfites that are included as antioxidants. Sensitivity to various sulfites is possible, especially among asthmatic or atopic patients. These principles are the basis for the flowchart presented in Figure 6. A patient should never be denied the benefit of local anesthesia based on flawed assumptions regarding allergy.
Managing patients allergic to local anesthetics. Rule out common reactions misinterpreted as allergy, eg, syncope and tachycardia. Then establish that the nature of their reaction at least resembled a hypersensitivity reaction, eg, rash, pruritus, urticaria, or dyspnea. If the drug is known, choose another amide, free of vasopressor so no sulfites are present. Otherwise refer the patient to an allergist, for testing of sulfites and exemplary local anesthetics such as lidocaine, mepivacaine, and prilocaine. (Adapted from deShazo and Kemp.)
Local Toxicity
Ischemic necrosis of tissues may follow injections of local anesthetics. This can be due to the irritating nature of a solution, pressure from large volumes, or constriction of the vasculature by vasopressors. This concern is greatest when injecting into attached mucosa such as the hard palate. There is also mounting concern regarding direct neurotoxicity related to formulations containing high concentrations such as 4% articaine and prilocaine.
Haas and Lennon reported an increased incidence of paresthesias in Canada following the introduction of articaine in the mid-1980s. In 1993 alone, 14 cases of paresthesia were reported, and all were attributed to articaine or prilocaine. When articaine was first submitted for approval to the Food and Drug Administration in the United States, it was identified as having a higher risk for paresthesia than lidocaine.
More recently, Garisto et al reviewed claims of paresthesia in the United States during the period of November 1997 through August 2008 and found 248 cases of paresthesia following dental procedures. Most cases (∼95%) involved mandibular nerve blocks, and in 89% of these the lingual nerve was affected. Compared to other local anesthetics, paresthesia was found to be 7.3 times more likely with 4% articaine and 3.6 times more likely with 4% prilocaine. Similar findings from reports of paresthesia in Denmark were published by Hillerup et al. This data may be even more significant when one considers the number of cases that may very well go unreported.
Although the dental community has been slow to reach consensus regarding this issue, it should be appreciated that the medical anesthesia literature is emphatic in claiming that greater concentration of local anesthetic solutions increases risk for direct neurotoxicity to nerve trunks: “All the clinically used local anesthetics can produce direct toxicity to nerves if they achieve sufficiently high intraneural concentrations. Clinicians should be aware that the concentrations of formulated local anesthetic solutions are neurotoxic per se and that their dilution, in situ or in tissue, is essential for safe use.”1
This fact is further supported by Hillerup et al, who demonstrated greater neural toxicity of 4 compared to 2% articaine in sciatic nerve preparations. As with all drugs, each practitioner needs to perform a risk-benefit analysis before using a medication. Only if the benefit of using articaine outweighs the risk for this practitioner in this patient should it be considered for use. It might be wise to limit the use of 4% concentrations for infiltration and avoid their use for nerve blocks, opting instead for agents formulated in lower concentrations.,
LOCAL ANESTHETIC COMPARISONS
Lidocaine continues its prominence as the most widely used local anesthetic in the United States, but all of these agents have comparable efficacy. They differ in potency and several pharmacokinetic parameters that account for differences in the onset and duration of anesthesia. Selection of a particular agent must take into account the duration of the procedure planned and issues regarding vasopressor concentrations. For lengthy procedures, bupivacaine is the logical choice, but it has been implicated as one of the more painful agents during injection according to studies that have compared various anesthetics.–, One strategy is to provide the initial 60–90 minutes of anesthesia using a less irritating agent (lidocaine or prilocaine) and then reinject the anesthetized tissue with bupivacaine to provide analgesia well into the postoperative period. Such a strategy is most effective following nerve blocks; shorter duration for pulpal anesthesia should be anticipated following infiltration. (See Table 2.)
Table 2.
Despite anecdotal claims regarding the superiority of articaine over lidocaine for inferior alveolar block, published studies have found little if any difference, especially when teeth are symptomatic.–, Any slight advantage of articaine is offset by its greater risk for paresthesia addressed above. However, for infiltration of the mandible, articaine is clearly superior and carries no risk for neural toxicity unless injected near the mental nerve.,, The superiority of articaine can be explained by its high lipid solubility and the concentration of its formulations. Unlike other anesthetics having benzene as their aromatic ring, articaine has a thiophene ring and substitutions that confer greater lipid solubility than other local anesthetics with the exception of bupivacaine. This property should have allowed its formulation in a lower concentration, but in fact it was formulated as a 4% solution. Not only is articaine more lipid soluble, but its formulation provides a greater number of molecules than an equal volume of 2% lidocaine, for example. To date there have been no published studies comparing articaine to 4% lidocaine solutions for mandibular infiltration. Lidocaine in this concentration would present an unacceptable risk for systemic toxicity, which introduces another attractive property of articaine: pattern of clearance.
Although articaine is classified as an amide, because of linkage of its intermediate chain, the thiophene ring also contains an ester side chain. This chain is hydrolyzed by plasma esterases rendering the molecule inactive. The result is that articaine has an elimination half-life of only 20–40 minutes compared to >90 minutes for lidocaine and other amides that require hepatic clearance. For this reason, articaine presents less risk for systemic toxicity during lengthy appointments when additional doses of anesthetic are administered. Be reminded, however, that a 4% concentration articaine contains twice the dose of 2% lidocaine per volume administered, and their maximum recommended doses are identical.
Maximum Doses for Local Anesthetics
Based on the data originally presented by Scott et al, lidocaine 400 mg injected submucosally produces systemic serum concentrations well below toxic levels. This is approximately the amount found in 10 dental anesthetic cartridges, and this number has been cited historically as the limit per dental appointment. Notwithstanding the fact that somewhat higher amounts can be used when formulated with vasopressors, this suggestion is obviously a safe guideline for lidocaine.
The elimination half-life (T1/2β) of the various local anesthetics ranges from 90 minutes for conventional agents such as lidocaine to >200 minutes for agents such as bupivacaine. This decline commences after peak serum concentration is achieved: approximately 20 minutes with anesthetics alone and ∼20–30 minutes for those combined with vasopressors., Once the peak concentration is achieved, additional doses will become absorbed as original doses are in decline. This is a perilous time because one cannot accurately predict the serum concentration at any period. Furthermore, patient responses follow a bell-shaped pattern of distribution and render these theoretical calculations even more problematic. Keep in mind that both liver and renal functions decline 50% by age 65 and beta blockers reduce hepatic blood flow. Articaine is the exception because it has an ester side chain and is inactivated in serum by plasma cholinesterases.
Frequently the dentist administers a combination of local anesthetic formulations, and it must be appreciated that systemic effects of these combinations follow principles of summation.1 When adhering to maximum dosage guidelines, systemic effects of various agents should be regarded as additive. For example, if you have administered half the maximum dose for lidocaine and wish to add bupivacaine, reduce its maximum dose by half.
VASOPRESSORS
Vasopressors are drugs that provide constriction of blood vessels by activating alpha-1 adrenergic receptors. They are combined with local anesthetics to provide hemostasis in the operative field and to delay anesthetic absorption. Delayed absorption of local anesthetics not only reduces the risk for systemic toxicity, but also prolongs the duration of anesthesia. Epinephrine is the most common agent used for this purpose, despite the fact that it exhibits considerable cardiac stimulation because of its additional action as a beta-1 adrenergic agonist.
Despite the popularity of epinephrine 1 ∶ 100,000, concentrations greater than 1 ∶ 200,000 (5 µg/mL) offer little if any advantage. Greater concentrations do not provide better onset or duration for inferior alveolar nerve block., Nor do higher concentrations reduce local anesthetic serum concentrations., However, greater concentrations, eg, 1 ∶ 100,000 (10 µg/mL) and 1 ∶ 50,000 (20 µg/mL), may provide better hemostasis when infiltrated at the surgical site when this influence is desired.
Cardiovascular Influences
To properly address safety issues, one must first appreciate principles of dosage calculations that were presented in Table 1 of this article. There is continued debate regarding deleterious influences of epinephrine on patients having cardiovascular disease. Often this dispute continues without fully appreciating the actual action and effects of this commonly used drug. Epinephrine acts as an agonist on alpha, beta-1, and beta-2 receptors. These actions account for its effects on the cardiovascular system, as illustrated in Figure 7. Before analyzing this figure, it is important to clarify a common misconception. The dentist regards epinephrine as a vasoconstrictor based on its effects when administered into submucosal tissues. This is because the tiny vessels in this location contain only alpha receptors. Larger systemic arteries that determine arterial resistance and diastolic blood pressure contain far more beta-2 than alpha receptors, and following absorption, low doses of epinephrine produce dilation of these vessels.
Cardiovascular effects of epinephrine.31 The following graph illustrates the typical cardiovascular response to epinephrine administered as a continuous intravenous infusion of 10 µg/min. (This is the amount contained in 1 mL of a 1 ∶ 100,000 concentration.) Epinephrine increases heart rate (HR) by activating beta-1 receptors in the sinoatrial node, the heart's normal pacemaker. It also activates beta-1 receptors on myocardial cells, increasing their contractility and increasing systolic blood pressure (SBP). However, it activates beta-2 receptors on systemic arteries producing vasodilation. This decline in arterial resistance produces a reduction in diastolic pressure (DBP). These effects result in little change of mean arterial pressure (MAP).
Clinical trials have confirmed unequivocally that even small dosages of epinephrine in local anesthetic solutions have an influence on cardiovascular function. Dionne et al studied the influence of 3 cartridges of 2% lidocaine with epinephrine 1 ∶ 100,000 (∼60 µg epinephrine). Submucosal injection of this dosage increased cardiac output, heart rate, and stroke volume. Systemic arterial resistance was reduced and mean arterial pressure remained essentially unchanged. Likewise, Hersh et al observed similar results following the administration of articaine containing 1 ∶ 100,000 (∼120 µg) and 1 ∶ 200,000 (∼60 µg) epinephrine, with a greater response from the higher dosage. These findings are consistent with well-established influences as illustrated in Figure 7.
The results of studies such as those just mentioned must be viewed in perspective. For example the influence of ∼120 µg epinephrine published by Hersh et al was minor: heart rate increases of ∼8–10 beats per minute and blood pressure changes of ∼5 mm Hg on average. However, the 14 participants were in perfect health, with resting vital signs that averaged 68 for heart rate, 125 mm Hg for systolic pressure, and 73 mm Hg for diastolic pressure. Furthermore, they were taking no significant medications. Obviously, such individuals can easily tolerate the dosages administered, but it should be noted that 2 of these healthy participants actually reported palpitations.
Even small doses of epinephrine produce cardiovascular effects; this is unequivocal. At issue is whether or not the cardiovascular influences of epinephrine pose a significant risk to patients having varying degrees of compromise. Standards and guidelines continue to be promoted, but in fact are all anecdotal. To suggest that a “2-cartridge” limit be imposed for patients with cardiovascular disease is naïve. Ultimately, the decision requires the dentist to exercise sound clinical judgment based on a thorough analysis of each patient under consideration. If consultation with the patient's physician is indicated, discuss the anticipated dosage range in terms of micrograms, not concentrations or cartridges. For example, if 2–4 cartridges of local anesthetic are planned, explain that you will be using 40–80 µg of epinephrine infiltrated submucosally, not 2–4 cartridges of epinephrine 1 ∶ 100,000. The physician is unfamiliar with a dosage expressed as cartridges or concentrations. As reference, consider the conventional epinephrine dose for managing an allergic reaction is 0.3 mg or 300 µg. A physician will generally be concerned with doses of 100 µg or greater.
Levonordefrin (NeoCobefrin) is the vasopressor combined with 2% mepivacaine formulations in the United States. It more closely resembles norepinephrine than epinephrine, lacking activity at beta-2 receptors. For this reason it elevates not only systolic blood pressure like epinephrine, but diastolic and mean arterial pressures as well. In some patients this can trigger a reflex vagal influence on heart rate that may offset some of its direct beta-1 receptor stimulation of heart rate. However, studies that assess cardiovascular influences following intraosseous injections have found little difference between epinephrine and levonordefrin., This likely is explained by the rapid absorption, which allows for direct beta-1 stimulation before reflex responses to mean arterial pressure intervene. A comparison of epinephrine and levonordefrin is presented in Table 3.
Table 3.
Maximum permissible doses of vasopressors have not been established. To express limits in terms of appointments is impractical; time of treatment may be as brief as 30 minutes or as long as 3–4 hours. Furthermore, the influence of a given dose of vasopressor among patients is highly variable. Peak influences of epinephrine are generally observed within 5–10 minutes following injection and they decline rapidly; epinephrine and levonordefrin are catecholamines and rapidly metabolized by catechol-o-methyltransferase. In fact, the elimination half-life for most catecholamines is only 1–3 minutes. Generally, the hemodynamic influences are witnessed within minutes of injection and have completely subsided in 10–15 minutes. An epinephrine dose of 40 µg (approximately 2 cartridges containing epinephrine 1 ∶ 100,000) is the most conservative and frequently cited dose limitation for patients having significant cardiovascular disease. It should be clarified that this guideline more appropriately reflects 30-minute time periods, not appointments. A more rational suggestion is to base the dosage on patient assessment, not maximal amounts. For example, if for any reason the medical status of a patient is in question, a sensible protocol is to record baseline heart rate and blood pressure preoperatively and again following every 20–40 µg administered. This would equate to 1–2 cartridges containing a 1 ∶ 100,000 epinephrine concentration. Virtually any patient can tolerate the cardiovascular influences of this amount. If the patient remains stable, additional doses may be administered and followed by a similar pattern of reassessing vital signs.
Drug Interactions
Potential drug interactions have been thoroughly addressed in a previous continuing education article in this journal. The most important of these relate to possible enhanced cardiovascular stimulation. Vasopressors found in local anesthetic formations have cardiotonic effects, and this may become more significant when patients are medicated with any drug having similar influences. These include tricyclic and monoamine oxidase inhibitor antidepressants, digoxin, thyroid hormone, or any of the sympathomimetics used for weight control or attention deficit disorders. Vasopressors are not contraindicated in these patients, but they should be administered with caution in a manner addressed above for medically compromised patients. For patients suspected of stimulant drug abuse, eg, cocaine, it may be wise to avoid vasopressors altogether.
Cautious use of vasopressors is also advised for patients medicated with nonselective beta blockers. Unlike selective agents that only block beta-1 receptors on the heart, nonselective agents also block vascular beta-2 receptors. In this case the alpha agonist action of vasopressors becomes more pronounced and both diastolic and mean arterial pressures can become dangerously elevated. This is generally accompanied by a sudden reflex slowing of heart rate. Significant consequences of this interaction are well documented.–, The interaction with beta blockers follows a time course identical to that observed for normal cardiovascular responses to epinephrine. It commences following absorption from the injection site, which generally peaks within 5 minutes and declines over the subsequent 10–15 minutes. Vasopressors are not contraindicated in patients taking nonselective beta blockers, but doses should be conservative and blood pressure monitored periodically during administration as described above. Gingival retraction cords impregnated with racemic epinephrine should be avoided. These products contain epinephrine in amounts far exceeding those contained in local anesthetic formulations.
LOCAL ANESTHETIC REVERSAL
In closing, it should be mentioned that a local anesthetic reversal agent has been introduced that effectively reverses the influence of vasopressors on submucosal vessels. Phentolamine (OraVerse) is an alpha receptor blocker formulated in dental cartridges. When it is injected into the identical site where anesthetic was administered, vessels dilate, leading to enhanced absorption of local anesthetic, which shortens the duration of anesthesia. It will likely receive limited use because of its expense and the fact that sustained anesthesia is generally a benefit during the postoperative period for pain management. However, it may be useful in the management of small children or patients with special needs who may be prone to self-inflected injury while tissues remain numb. A consideration may also be given to the fragile diabetic or elderly patient for whom adequate nutritional intake may be hindered by prolonged numbness. Reversal may also be offered to the busy patient who must return to work and communicate effectively.
CONTINUING EDUCATION QUESTIONS
- 1. All of the following influence duration of local anesthesia EXCEPT:
- A. Addition of vasopressor to the formulation
- B. Dissociation constant (pKa) of the local anesthetic
- C. Relative protein binding affinity of the local anesthetic
- D. Relative vasodilating property of the local anesthetic
- 2. The maximum recommended dose for lidocaine with epinephrine is 500 mg, and 90 mg for bupivacaine. Anesthesia is difficult to obtain and you have administered 6 cartridges of 2% lidocaine with epinephrine to remove 4 third molars. The lower molars remain sensitive and you elect to reinject using 0.5% bupivacaine 1 ∶ 200,000 epinephrine. Which of the following number of cartridges would be the maximum number you can safely administer? (Assume 2 mL per cartridge.)
- A. 2–3
- B. 4–5
- C. 8–9
- D. 10–11
- 3. The risk for direct neurotoxicity from local anesthetics is most closely associated with which of the following characteristics?
- A. Greater concentration
- B. Greater lipid solubility
- C. Higher pH
- D. Lower pKa
- 4. Which of the following is the most common initial life-threatening consequence of local anesthetic overdose?
- A. Anaphylactic reactions
- B. Convulsive seizure
- C. Respiratory arrest
- D. Ventricular fibrillation
References
1. Berde CB, Strichartz GR. Local anesthetics. In: Miller RD, Eriksson LI, Fleisher LA, et al., editors. Miller's Anesthesia. 7th ed. Philadelphia, Pa: Elsevier, Churchill Livingstone; 2009. [Google Scholar]
2. Katzung BG, White PF. Local anesthetics. In: Katzung BG, Masters SB, Trevor AJ, editors. Basic and Clinical Pharmacology. 11th ed. New York, NY: McGraw-Hill Companies Inc; 2009. [Google Scholar]
3. Goodson JM, Moore PA. Life-threatening reactions after pedodontic sedation: an assessment of narcotic, local anesthetic and antiemetic drug interactions. J Am Dent Assoc. 1983;107:239–245. [PubMed] [Google Scholar]
4. Scott DB, Jebson PJR, Braid DP, et al. Factors affecting plasma levels of lignocaine and prilocaine. Brit J Anaesth. 1972;44:1040–1049. [PubMed] [Google Scholar]
5. Hersh EV, Giannakopoulos H, Levin LM, et al. The pharmacokinetics and cardiovascular effects of high-dose articaine with 1 ∶ 100,000 and 1 ∶ 200,000 epinephrine. J Am Dent Assoc. 2006;137:1562–1571. [PubMed] [Google Scholar]
6. Benz EJ. Disorders of hemoglobin. In: Longo DL, Kasper DL, Jameson JL, et al., editors. Harrison's Principles of Internal Medicine. 18th ed. New York, NY: McGraw Hill; 2012. [Google Scholar]
7. Schatz M. Adverse reactions to local anesthetics. Immunol Allergy Clin North Am. 1992;12:585–609.[Google Scholar]
8. Gell PGH, Coombs RRA. Classification of allergic reactions responsible for clinical hypersensitivity and disease. In: Gell PGH, Coombs RRA, Hachmann PJ, editors. Clinical Aspects of Immunology. 3rd ed. Oxford, England: Blackwell Scientific; 1975. [Google Scholar]
9. Adkinson NF., Jr . Drug allergy. In: Adkinson NF Jr, Yunginger JW, Busse WW, et al., editors. Middleton's Allergy: Principles and Practice. 6th ed. Philadelphia, Pa: Mosby Inc; 2003. [Google Scholar]
10. Gall H, Kaufmann R, Kalveram CM. Adverse reactions to local anesthetics: analysis of 197 cases. J Allergy Clin Immunol. 1996;97:933–937. [PubMed] [Google Scholar]
11. Berkun Y, Ben-Zvi A, Levy Y, Galili D, Shalit M. Evaluation of adverse reactions to local anesthetics: experience with 236 patients. Ann Allergy Asthma Immunol. 2003;91:342–345. [PubMed] [Google Scholar]
12. Speca SJ, Boynes SG, Cuddy MA. Allergic reactions to local anesthetic formulations. Dent Clin North Am. 2010;54:655–664. [PubMed] [Google Scholar]
13. deShazo RD, Kemp SF. Allergic reactions to drugs and biologic agents. JAMA. 1997;278:1895–1906. [PubMed] [Google Scholar]
14. Haas DA, Lennon D. A 21 year retrospective study of reports of paresthesia following local anesthetic administration. J Can Dent Assoc. 1995;61:319–330. [PubMed] [Google Scholar]
15. Garisto GA, Gaffen AS, Lawrence HP, Tenenbaum HC, Haas DA. Occurrence of paresthesia after dental local anesthetic administration in the United States. J Am Dent Assoc. 2010;141:836–844. [PubMed] [Google Scholar]
16. Hillerup S, Jensen RH, Ersboll BK. Trigeminal nerve injury associated with injection of local anesthetics: needle lesion or neurotoxicity. J Am Dent Assoc. 2011;142:531–539. [PubMed] [Google Scholar]
17. Hillerup S, Bakke M, Larsen JO, Thomsen CE, Gerds TA. Concentration-dependent neurotoxicity of articaine: an electrophysiological and stereological study of the rat sciatic nerve. Anesth Analg. 2011;112:1330–1338. [PubMed] [Google Scholar]
18. Morris R, McKay W, Mushlin P. Comparison of pain associated with intradermal and subcutaneous infiltration with various local anesthetic solutions. Anesth Analg. 1987;66:1180–1182. [PubMed] [Google Scholar]
19. Wahl MJ, Overton D, Howell J, Siegel E, Schmitt MM, Muldoon M. Pain on injection of prilocaine plain vs. lidocaine with epinephrine. A prospective double-blind study. J Am Dent Assoc. 2001;132:1396–1401. [PubMed] [Google Scholar]
20. Wahl MJ, Schmitt MM, Overton DA, Gordon MK. Injection pain of bupivacaine with epinephrine vs. prilocaine plain. J Am Dent Assoc. 2002;133:1652–1656. [PubMed] [Google Scholar]
21. Yagiela JA. Local anesthetics. In: Dionne RA, Phero JP, Becker DE, editors. Management of Pain & Anxiety in the Dental Office. St Louis, Mo: WB Saunders/Elsevier Science; 2002. [Google Scholar]
22. Brandt RG, Anderson PF, McDonald NJ, Sohn W, Peters MC. The pulpal anesthetic efficacy of articaine versus lidocaine in dentistry: a meta-analysis. J Am Dent Assoc. 2011;142:493–504. [PubMed] [Google Scholar]
23. Malamed SF, Gagnon S, Leblanc D. Efficacy of articaine: a new amide local anesthetic. J Am Dent Assoc. 2000;131:635–642. [PubMed] [Google Scholar]
24. Mikesell P, Nusstein J, Reader A, Beck M, Weaver J. A comparison of articaine and lidocaine for inferior alveolar nerve blocks. J Endod. 2005;31:265–270. [PubMed] [Google Scholar]
25. Robertson D, Nusstein J, Reader A, Beck M, McCartney M. The anesthetic efficacy of articaine in buccal infiltration of mandibular posterior teeth. J Am Dent Assoc. 2007;138:1104–1112. [PubMed] [Google Scholar]
26. Abdulwahab M, Boynes S, Moore P, et al. The efficacy of six local anesthetic formulations used for posterior mandibular buccal infiltration anesthesia. J Am Dent Assoc. 2009;140:1018–1024. [PubMed] [Google Scholar]
27. Moore PA, Hersh EV, Papas AS, et al. Pharmacokinetics of lidocaine with epinephrine following local anesthesia reversal with phentolamine mesylate. Anesth Prog. 2008;55:40–48.[PMC free article] [PubMed] [Google Scholar]
28. Montamat SC, Cusack BJ, Vestal RE. Management of drug therapy in the elderly. N Engl J Med. 1989;321:303–309. [PubMed] [Google Scholar]
29. Dagher FB, Yared GM, Machtou P. An evaluation of 2% lidocaine with different concentrations of epinephrine for inferior alveolar nerve block. J Endod. 1997;23:178–180. [PubMed] [Google Scholar]
30. Tofoli GR, Ramacciato JC, de Oliveira PC, et al. Comparison of effectiveness of 4% articaine associated with 1 ∶ 100,000 or 1 ∶ 200,000 epinephrine in inferior alveolar nerve block. Anesth Prog. 2003;50:164–168.[PMC free article] [PubMed] [Google Scholar]
31. Westfall TC, Westfall DP. Adrenergic agonists and antagonists. In: Brunton LL, Chabner BA, Knollmann BC, editors. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 12th ed. New York, NY: McGraw-Hill Companies Inc; 2011. [Google Scholar]
32. Dionne RA, Goldstein DS, Wirdzek PR. Effects of diazepam premedication and epinephrine-containing local anesthetic on cardiovascular and catecholamine responses to oral surgery. Anesth Analg. 1984;63:640–646. [PubMed] [Google Scholar]
33. Guglielmo A, Reader A, Nist R, Beck M, Weaver J. Anesthetic efficacy and heart rate effects of the supplemental intraosseous injection of 2% mepivacaine with 1 ∶ 20,000 levonordefrin. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;87:284–293. [PubMed] [Google Scholar]
34. Lawaty I, Drum M, Reader A, Nusstein J. A prospective, randomized, double-blind comparison of 2% mepivacaine with 1 ∶ 20,000 levonordefrin versus 2% lidocaine with 1 ∶ 100,000 epinephrine for maxillary infiltrations. Anesth Prog. 2010;57:139–144.[PMC free article] [PubMed] [Google Scholar]
35. Becker DE. Adverse drug interactions. Anesth Prog. 2011;58:31–41.[PMC free article] [PubMed] [Google Scholar]
36. Foster CA. Aston SJ. Propranolol-epinephrine interaction: a potential disaster. Plast Reconstr Surg. 1983;72:74–78. [PubMed] [Google Scholar]
37. Gandy W. Severe epinephrine-propranolol interaction. Ann Emerg Med. 1989;18:98–99. [PubMed] [Google Scholar]
38. Mito RS, Yagiela JA. Hypertensive response to levonordefrin in a patient receiving propranolol: report of a case. J Am Dent Assoc. 1988;116:55–57. [PubMed] [Google Scholar]
Articles from Anesthesia Progress are provided here courtesy of American Dental Society of Anesthesiology
Many local anesthetics fall into two general chemical classes, amino esters (top) and amino amides (bottom)
A local anesthetic (LA) is a medication that causes absence of pain sensation. When it is used on specific nerve pathways (local anesthetic nerve block), paralysis (loss of muscle power) also can be achieved.
Clinical LAs belong to one of two classes: aminoamide and aminoester local anesthetics. Synthetic LAs are structurally related to cocaine. They differ from cocaine mainly in that they have a very low abuse potential and do not produce hypertension or (with few exceptions) vasoconstriction.
They are used in various techniques of local anesthesia such as:
- Topical anesthesia (surface)
- Topical administration of cream, gel, ointment, liquid, or spray of anaesthetic dissolved in DMSO or other solvents/carriers for deeper absorption
- Spinal anesthesia (subarachnoid block)
- 1Medical uses
- 3Side effects
- 3.1Localized side effects
- 3.2Potential side effects
- 5Techniques
- 6Types
Medical uses[edit]
Acute pain[edit]
Acute pain may occur due to trauma, surgery, infection, disruption of blood circulation, or many other conditions in which tissue injury occurs. In a medical setting, pain alleviation is desired when its warning function is no longer needed. Besides improving patient comfort, pain therapy can also reduce harmful physiological consequences of untreated pain.
Acute pain can often be managed using analgesics. However, conduction anesthesia may be preferable because of superior pain control and fewer side effects. For purposes of pain therapy, LA drugs are often given by repeated injection or continuous infusion through a catheter. LA drugs are also often combined with other agents such as opioids for synergistic analgesic action.[1] Low doses of LA drugs can be sufficient so that muscle weakness does not occur and patients may be mobilized.
Some typical uses of conduction anesthesia for acute pain are:
- Labor pain (epidural anesthesia, pudendal nerve blocks)
- Postoperative pain (peripheral nerve blocks, epidural anesthesia)
- Trauma (peripheral nerve blocks, intravenous regional anesthesia, epidural anesthesia)
Chronic pain[edit]
Chronic pain is a complex and often serious condition that requires diagnosis and treatment by an expert in pain medicine. LAs can be applied repeatedly or continuously for prolonged periods to relieve chronic pain, usually in combination with medication such as opioids, NSAIDs, and anticonvulsants. Though it can be easily performed, repeated local anaesthetic blocks in chronic pain conditions are not recommended as there is no evidence of long-term benefits.[2]
Surgery[edit]
Virtually every part of the body can be anesthetized using conduction anesthesia. However, only a limited number of techniques are in common clinical use. Sometimes, conduction anesthesia is combined with general anesthesia or sedation for the patient's comfort and ease of surgery. However, many anaesthetists, surgeons, patients and nurses believe that it is safer to perform major surgeries under local anesthesia than general anesthesia.[3] Typical operations performed under conduction anesthesia include:
- Dentistry (surface anesthesia, infiltration anesthesia or intraligamentary anesthesia during restorative operations such as fillings, crowns, and root canals,[4] or extractions, and regional nerve blocks during extractions and surgeries)
- Podiatry (cutaneous, nail avulsions, matricectomy, bunionectomy, hammertoe repair [4] and various other podiatric procedures)
- Eye surgery (surface anesthesia with topical anesthetics or retrobulbar block during cataract removal or other ophthalmic procedures[4])
- ENT operations, head and neck surgery (infiltration anesthesia, field blocks, or peripheral nerve blocks, plexus anesthesia)
- Shoulder and arm surgery (plexus anesthesia or intravenous regional anesthesia)[5]
- Heart and lung surgery (epidural anesthesia combined with general anesthesia)
- Abdominal surgery (epidural anesthesia/spinal anesthesia, often combined with general anesthesia during inguinal hernia repair or other abdominal surgery [4])
- Gynecological, obstetrical, and urological operations (spinal/epidural anesthesia)
- Bone and joint surgery of the pelvis, hip, and leg (spinal/epidural anesthesia, peripheral nerve blocks, or intravenous regional anesthesia)
- Surgery of skin and peripheral blood vessels (topical anesthesia, field blocks, peripheral nerve blocks, or spinal/epidural anesthesia)
Diagnostic Tests[edit]
Diagnostic tests such as bone marrow aspiration, lumbar puncture (spinal tap) and aspiration of cysts or other structures are made to be less painful upon administration of local anesthetic before insertion of larger needles.[4]
Other uses[edit]
Local anesthesia is also used during insertion of IV devices, such as pacemakers and implantable defibrillators, ports used for giving chemotherapy medications and hemodialysis access catheters.[4]
Topical anesthesia, in the form of lidocaine/prilocaine (EMLA) is most commonly used to enable relatively painless venipuncture (blood collection) and placement of intravenous cannulae. It may also be suitable for other kinds of punctures such as ascites drainage and amniocentesis.
Surface anesthesia also facilitates some endoscopic procedures such as bronchoscopy (visualization of the lower airways) or cystoscopy (visualization of the inner surface of the bladder).
Properties of Ideal Anesthetic[edit]
- It should not irritate the tissue to which it is applied.
- It should not make any long-lasting changes on nerve structure.
- Its systemic toxicity should be minimal.
- It must be effective regardless of whether it is injected into tissue or applied locally on mucous membranes.
- The time of onset of anesthesia should be minimal.
- Duration of action must be sufficiently long to allow the procedure to be completed but not so long as to necessitate extended recovery.
- It should have enough potency to administer full anesthesia without supplementing additional concentrated solutions that are potentially damaging.
- It should not produce allergic reaction.
- It should be stable in solution and should spontaneously undergo biotransformation in the body.
- It should be sterile or capable of being sterilized by heat without deterioration.[6]
Side effects[edit]
Localized side effects[edit]
Edema of tongue, pharynx and larynx may develop as a side effect of local anaesthesia. This could be caused by a variety of reasons including trauma during injection, infection, an allergic reaction, haematoma or injection of irritating solutions such as cold-sterilisation solutions. Usually there is tissue swelling at the point of injection. This is due to puncturing of the vein which allows the blood to flow into loose tissues in the surrounding area. Blanching of the tissues in the area where the local anaesthetic is deposited is also common. This gives the area a white appearance as the blood flow is prevented due to vasoconstriction of arteries in the area. The vasoconstriction stimulus gradually wears off and subsequently the tissue returns to normal in less than 2 hours.[7]
The side effects of inferior alveolar nerve block include feeling tense, clenching of the fists and moaning.[8]
The duration of soft tissue anaesthesia is longer than pulpal anaesthesia and is often associated with difficulty eating, drinking and speaking.[8]
Risks[edit]
The risk of temporary or permanent nerve damage varies between different locations and types of nerve blocks.[9]
There is risk of accidental damage to local blood vessels during injection of the local anaesthetic solution. This is referred to as Haematoma and could result in pain, trismus, swelling and/or discolouration of the region. The density of tissues surrounding the injured vessels is an important factor for Haematoma. There is greatest chance of this occurring in a posterior superior alveolar nerve block or in a pterygomandibular block.
Giving local anaesthesia to patients with liver disease can have significant consequences. Thorough evaluation of the disease should be carried out to assess potential risk to the patient as in significant liver dysfunction, the half-life of amide local anaesthetic agents may be drastically increased thus increasing the risk of overdose.
Local anaesthetics and vasoconstrictors may be administered to pregnant patients however it is very important to be extra cautious when giving a pregnant patient any type of drug. Lidocaine can be safely used but bupivacaine and mepivacaine should be avoided. Consultation with the obstetrician is vital before administrating any type of local anaesthetic to a pregnant patient.[7]
Recovery[edit]
Permanent nerve damage after a peripheral nerve block is rare. Symptoms are likely to resolve within a few weeks. The vast majority of those affected (92%–97%) recover within four to six weeks; 99% of these people have recovered within a year. An estimated one in 5,000 to 30,000 nerve blocks results in some degree of permanent persistent nerve damage.[9]
Symptoms may continue to improve for up to 18 months following injury.
Potential side effects[edit]
General systemic adverse effects are due to the pharmacological effects of the anesthetic agents used. The conduction of electric impulses follows a similar mechanism in peripheral nerves, the central nervous system, and the heart. The effects of local anesthetics are, therefore, not specific for the signal conduction in peripheral nerves. Side effects on the central nervous system and the heart may be severe and potentially fatal. However, toxicity usually occurs only at plasma levels which are rarely reached if proper anesthetic techniques are adhered to. High plasma levels might arise, for example, when doses intended for epidural or intrasupport tissue administration are accidentally delivered as intravascular injection.[citation needed]
Emotional reactions[edit]
When patients are emotionally affected in the form of nervousness or fear, it can lead to vasovagal collapse. This is the anticipation of pain during administration that activates the parasympathetic nervous system while inhibiting the orthosympathetic nervous system.[10] What results is a dilation of arteries in muscles which can lead to a reduction in circulating blood volume inducing a temporary shortness of blood flow to the brain. Notable symptoms include restlessness, visibly looking pale, perspiration and possible the loss of consciousness. In severe cases, clonic cramps resembling an epileptic insult may occur.[10]
On the other hand, fear of administration can also result in accelerated, shallow breathing, or hyperventilation. The patient may feel a tingling sensation in hands and feet or a sense of light-headedness and increased chest pressure.
Hence, it is crucial for the medical professional administrating the local anaesthesia, especially in the form of an injection, to ensure that the patient is in a comfortable setting and has any potential fears alleviated in order to avoid these possible complications.
Central nervous system[edit]
Depending on local tissue concentrations of local anesthetics, excitatory or depressant effects on the central nervous system may occur.
Initial symptoms of systemic toxicity include ringing in the ears (tinnitus), a metallic taste in the mouth, tingling or numbness of the mouth, dizziness and/or disorientation.
At higher concentrations, a relatively selective depression of inhibitory neurons results in cerebral excitation, which may lead to more advanced symptoms include motor twitching in the periphery followed by grand mal seizures. It is reported that seizures are more likely to occur when bupivacaine is used, particularly in combination with chloroprocaine.[11]
A profound depression of brain functions may occur at even higher concentrations which may lead to coma, respiratory arrest, and death.[12] Such tissue concentrations may be due to very high plasma levels after intravenous injection of a large dose.
Another possibility is direct exposure of the central nervous system through the cerebrospinal fluid, i.e., overdose in spinal anesthesia or accidental injection into the subarachnoid space in epidural anesthesia.
Cardiovascular system[edit]
Cardiac toxicity can result from improper injection of agent into a vessel. Even with proper administration, it is inevitable for some diffusion of agent into the body from the site of application due to unforeseeable anatomical idiosyncrasies of the patient.[11] This may affect the nervous system or cause the agent to enter into general circulation. However, infections are very seldom transmitted.
Cardiac toxicity associated with overdose of intravascular injection of local anesthetic is characterized by hypotension, atrioventricular conduction delay, idioventricular rhythms, and eventual cardiovascular collapse. Although all local anesthetics potentially shorten the myocardial refractory period, bupivacaine blocks the cardiac sodium channels, thereby making it most likely to precipitate malignant arrhythmias. Even levobupivacaine and ropivacaine (single-enantiomer derivatives), developed to ameliorate cardiovascular side effects, still harbor the potential to disrupt cardiac function.[13] Toxicity from anesthetic combinations is additive.[11]
Endocrine[edit]
Endocrine and metabolic systems only have slightly adverse effects with most cases being without clinical repercussions.[11]
Immunologic allergy[edit]
Adverse reactions to local anesthetics (especially the esters) are not uncommon, but legitimate allergies are very rare. Allergic reactions to the esters is usually due to a sensitivity to their metabolite, para-aminobenzoic acid, and does not result in cross-allergy to amides.[14][15] Therefore, amides can be used as alternatives in those patients. Nonallergic reactions may resemble allergy in their manifestations. In some cases, skin tests and provocative challenge may be necessary to establish a diagnosis of allergy. Also cases of allergy to paraben derivatives occur, which are often added as preservatives to local anesthetic solutions.
Methemoglobinemia[edit]
Methemoglobinemia is a process where iron in hemoglobin is altered, reducing its oxygen-carrying capability, which produces cyanosis and symptoms of hypoxia. Exposure to aniline group chemicals such as benzocaine, lidocaine, and prilocaine can produce this effect, especially benzocaine.[14][15] The systemic toxicity of prilocaine is comparatively low, but its metabolite, o-toluidine, is known to cause methemoglobinemia.
Second-generation effects[edit]
Application of local anesthetics during oocyte removal during in vitro fertilisation has been up to debate. Pharmacological concentrations of anesthetic agents have been found in follicular fluid.[11] Clinical trials have not concluded any effects on pregnant women. However, there is some concern with the behavioral effects of lidocaine on offspring in rats.[11]
During pregnancy, it is not common for local anesthetics to have any adverse effect on the fetus. Despite this, risks of toxicity may be higher in pregnancy due to an increase in unbound fraction of local anesthetic and physiological changes increase the transfer of local anesthetic into the central nervous system.[11] Hence, it is recommended that pregnant women use a lower dose of local anesthetic to reduce any potential complications.
Treatment of overdose: 'Lipid rescue'[edit]
This method of toxicity treatment was invented by Dr. Guy Weinberg in 1998, and was not widely used until after the first published successful rescue in 2006. Evidence indicates Intralipid, a commonly available intravenous lipid emulsion, can be effective in treating severe cardiotoxicity secondary to local anesthetic overdose, including human case reports of successful use in this way (lipid rescue).[16][17][18][19][20] However, the evidence at this point is still limited.[21]
Though most reports to date have used Intralipid, a commonly available intravenous lipid emulsion, other emulsions, such as Liposyn and Medialipid, have also been shown effective.
Ample supporting animal evidence[16][17] and human case reports show successful use in this way.[19][20] In the UK, efforts have been made to publicise this use more widely[18] and lipid rescue has now been officially promoted as a treatment by the Association of Anaesthetists of Great Britain and Ireland.[22] One published case has been reported of successful treatment of refractory cardiac arrest in bupropion and lamotrigine overdose using lipid emulsion.[23]
The design of a 'homemade' lipid rescue kit has been described.[24]
Although lipid rescue mechanism of action is not completely understood, the added lipid in the blood stream may act as a sink, allowing for the removal of lipophilic toxins from affected tissues. This theory is compatible with two studies on lipid rescue for clomipramine toxicity in rabbits[25][26] and with a clinical report on the use of lipid rescue in veterinary medicine to treat a puppy with moxidectin toxicosis.[27]
Mechanism of action[edit]
All LAs are membrane-stabilizing drugs; they reversibly decrease the rate of depolarization and repolarization of excitable membranes (like nociceptors). Though many other drugs also have membrane-stabilizing properties, not all are used as LAs (propranolol, for example, though it has LA properties).LA drugs act mainly by inhibiting sodium influx through sodium-specific ion channels in the neuronalcell membrane, in particular the so-called voltage-gated sodium channels. When the influx of sodium is interrupted, an action potential cannot arise and signal conduction is inhibited. The receptor site is thought to be located at the cytoplasmic (inner) portion of the sodium channel. Local anesthetic drugs bind more readily to sodium channels in an activated state, thus onset of neuronal blockade is faster in rapidly firing neurons. This is referred to as state-dependent blockade.
LAs are weak bases and are usually formulated as the hydrochloride salt to render them water-soluble. At a pH equal to the protonated base's pKa, the protonated (ionized) and unprotonated (unionized) forms of the molecule exist in equimolar amounts, but only the unprotonated base diffuses readily across cell membranes. Once inside the cell, the local anesthetic will be in equilibrium, with the formation of the protonated (ionized) form, which does not readily pass back out of the cell. This is referred to as 'ion-trapping'. In the protonated form, the molecule binds to the LA binding site on the inside of the ion channel near the cytoplasmic end. Most LAs work on the internal surface of the membrane - the drug has to penetrate the cell membrane, which is achieved best in the nonionised form.
Acidosis such as caused by inflammation at a wound partly reduces the action of LAs. This is partly because most of the anesthetic is ionized and therefore unable to cross the cell membrane to reach its cytoplasmic-facing site of action on the sodium channel.
All nerve fibers are sensitive to LAs, but due to a combination of diameter and myelination, fibers have different sensitivities to LA blockade, termed differential blockade. Type B fibers (sympathetic tone) are the most sensitive followed by type C (pain), type A delta (temperature), type A gamma (proprioception), type A beta (sensory touch and pressure), and type A alpha (motor). Although type B fibers are thicker than type C fibers, they are myelinated, thus are blocked before the unmyelinated, thin C fiber.[medical citation needed][28]
Techniques[edit]
Local anesthetics can block almost every nerve between the peripheral nerve endings and the central nervous system. The most peripheral technique is topical anesthesia to the skin or other body surface. Small and large peripheral nerves can be anesthetized individually (peripheral nerve block) or in anatomic nerve bundles (plexus anesthesia). Spinal anesthesia and epidural anesthesia merge into the central nervous system.
Injection of LAs is often painful. A number of methods can be used to decrease this pain, including buffering of the solution with bicarbonate and warming.[29]
Clinical techniques include:
- Surface anesthesia is the application of an LA spray, solution, or cream to the skin or a mucous membrane; the effect is short lasting and is limited to the area of contact.
- Infiltration anesthesia is infiltration of LA into the tissue to be anesthetized; surface and infiltration anesthesia are collectively topical anesthesia
- Field block is subcutaneous injection of an LA in an area bordering on the field to be anesthetized.
- Peripheral nerve block is injection of LA in the vicinity of a peripheral nerve to anesthetize that nerve's area of innervation.
- Plexus anesthesia is injection of LA in the vicinity of a nerve plexus, often inside a tissue compartment that limits the diffusion of the drug away from the intended site of action. The anesthetic effect extends to the innervation areas of several or all nerves stemming from the plexus.
- Epidural anesthesia is an LA injected into the epidural space, where it acts primarily on the spinal nerve roots; depending on the site of injection and the volume injected, the anesthetized area varies from limited areas of the abdomen or chest to large regions of the body.
- Spinal anesthesia is an LA injected into the cerebrospinal fluid, usually at the lumbar spine (in the lower back), where it acts on spinal nerve roots and part of the spinal cord; the resulting anesthesia usually extends from the legs to the abdomen or chest.
- Intravenous regional anesthesia (Bier's block) is when blood circulation of a limb is interrupted using a tourniquet (a device similar to a blood-pressure cuff), then a large volume of LA is injected into a peripheral vein. The drug fills the limb's venous system and diffuses into tissues, where peripheral nerves and nerve endings are anesthetized. The anesthetic effect is limited to the area that is excluded from blood circulation and resolves quickly once circulation is restored.
- Local anesthesia of body cavities includes intrapleural anesthesia and intra-articular anesthesia.
- Transincision (or transwound) catheter anesthesia uses a multilumen catheter inserted through an insicion or wound and aligned across it on the inside as the incision or wound is closed, providing continuous administration of local anesthetic along the incision or wounds[30]
Dental-specific techniques include:
Vazirani-Alkinosi Technique[edit]
Cannot save large file on micro sd. The Vazirani-alkinosi technique is also known as the closed-mouth mandibular nerve block. It is mostly used in patients who have limited opening of the mandible or in those that have trismus; spasm of the muscles of mastication. The nerves which are anesthetised in this technique are the inferior alveolar, incisive, mental, lingual and mylohyoid nerves.
Dental needles are available in 2 lengths; short and long. As Vazirani-akinosi is a local anaesthetic technique which requires penetration of a significant thickness of soft tissues, a long needle is used. The needle is inserted into the soft tissue which covers the medial border of the mandibular ramus, in region of the inferior alveolar, lingual and mylohyoid nerves. The positioning of the bevel of the needle is very important as it must be positioned away from the bone of the mandibular ramus and instead towards the midline.[6]
Intraligamentary Infiltration[edit]
Intraligamentary infiltration, also known as periodontal ligament injection or intraligamentary injection (ILIs), is known as “the most universal of the supplemental injections”. ILIs are usually administered when inferior alveolar nerve block techniques are inadequate or ineffective.[31] ILIs are purposed for:
1. Single-tooth anesthesia
2. Low anesthetic dose
3. Contraindication for systemic anesthesia
4. Presence of systemic health problems[32]
ILI utilization is expected to increase because dental patients prefer fewer soft tissue anesthesia and dentists aim to reduce administration of traditional inferior alveolar nerve block (INAB) for routine restorative procedures.[33]
Injection methodology: The periodontal ligament space provides an accessible route to the cancellous alveolar bone, and the anesthetic reaches the pulpal nerve via natural perforation of intraoral bone tissue.[34][35]
Advantages of ILI over INAB: rapid onset (within 30 seconds), small dosage required (0.2-1.0mL), limited area of numbness,[36][37] lower intrinsic risks such as neuropathy, hematoma, trismus/jaw sprain[38][39][39] and self-inflicted periodontal tissue injury,[40][41] as well as decreased cardiovascular disturbances.[42] Its usage as a secondary or supplementary anesthesia on the mandible has reported a high success rate of above 90%.[43][44]
Disadvantages: Risk of temporary periodontal tissue damage, likelihood of bacteriemia and endocarditis for at-risk populations,[45] appropriate pressure and correct needle placement are imperative for anesthetic success, short duration of pulpal anesthesia limits the use of ILIs for several restorative procedures that require longer duration,[45] postoperative discomfort, and injury on unerupted teeth such as enamel hypoplasia and defects.
Technique description:
- All plaque and calculus to be eradicated, optimally before the operative visit to assist gingival tissue healing.
- Before injection, disinfect gingival sulcus with 0.2% chlorhexidine solution.[46]
- Administration of soft tissue anesthesia is recommended prior to ILI administration. This helps to enhance patient comfort.
- Needle gauges of sizes 27-gauge short or 30-gauge ultra-short needle are usually utilized.[47]
- The needle is inserted along the long axis, at a 30-degree angle, of the mesial or distal root for single rooted teeth and on the mesial and distal roots of multi-rooted teeth. Bevel orientation toward the root provides easier advancement of the needle apically.[48]
- When the needle reaches between the root and crestal bone, significant resistance is experience.
- Anesthetic deposition is recommended at 0.2mL, per root or site, over minimally 20 seconds.
- For its success, the anesthetic must be administered under pressure. It must not leak out of the sulcus into the mouth.
- Withdraw needle for minimally 10–15 seconds to permit complete deposition of solution. This can be slower than other injections as there is pressure build-up from the anesthetic administration.
- Blanching of the tissue is observed and may be more evident when vasoconstrictors are used. It is caused by a temporary obstruction of blood flow to the tissue.[48]
Syringes:
- Standard syringes can be used.
- The intraligamentary syringe offers mechanical advantage by using a trigger-grasp or click apparatus to employ a gear or lever that improves control and results in increased force to push the anesthetic cartridge's rubber stopper forward for medication deposition with greater ease.
- C-CLADs (computer controlled local anesthetic delivery devices) can be used. Its usage of computer microprocessors allows for control of fluid dynamics and anesthetic deposition. This minimizes subjective flow rates and variability in pressure. This thereby results in enhanced hydrodynamic diffusion of solution into bone or the target area of deposition,[49][50] thus permitting larger amounts of anesthetic solution to be delivered during ILIs without increased tissue damage.[51][52][53]
Things to note:
- ILIs are not recommended for patients with active periodontal inflammation.
- ILIs should not be administered at tooth sites with 5mm or more of periodontal attachment loss.
Gow-Gates Technique[edit]
Gow-Gates technique is used to provide anesthetics to the mandible of the patient's mouth. With the aid of extra and intraoral landmarks, the needle is injected into the intraoral latero-anterior surface of the condyle, steering clear below the insertion of the lateral pterygoid muscle.[54] The extraoral landmarks used for this technique are the lower border of the ear tragus, corners of the mouth and the angulation of the tragus on the side of the face.[54]
Biophysical forces (pulsation of the maxillary artery, muscular function of jaw movement) and gravity will aid with the diffusion of anesthetic to fill the whole pterygomandibular space. All three oral sensory parts of the mandibular branch of the trigeminal nerve and other sensory nerves in the region will come in contact with the anesthetic and this reduces the need to anesthetise supplementary innervation.[54]
In comparison to other regional block methods of anestheising the lower jaw, the Gow-Gates technique has a higher success rate in fully anesthetising the lower jaw. One study found that out of 1,200 patients receiving injections through the Gow-Gate technique, only 2 of them did not obtain complete anesthesia.[54]
Types[edit]
This LA system is designed to prevent needlestick injury. A cartridge of LA fits into the disposable needle, which can be locked when not in use and can be separated from the handle.
Local anesthetic solutions for injection typically consist of:[55]
- The local anesthetic agent itself
- A vehicle, which is usually water-based or just sterile water
- Vasoconstrictor possibly (see below)
- Reducing agent (antioxidant), e.g. if epinephrine is used, then sodium metabisulfite is used as a reducing agent
- Preservative, e.g. methylparaben
Esters are prone to producing allergic reactions, which may necessitate the use of an amide. The names of each locally clinical anesthetic have the suffix '-caine'.Most ester LAs are metabolized by pseudocholinesterase, while amide LAs are metabolized in the liver. This can be a factor in choosing an agent in patients with liver failure,[56] although since cholinesterases are produced in the liver, physiologically (e.g. very young or very old individual) or pathologically (e.g. cirrhosis) impaired hepatic metabolism is also a consideration when using esters.
Sometimes, LAs are combined, e.g.:
- Lidocaine/prilocaine (EMLA, eutectic mixture of local anesthetic)
- Lidocaine/tetracaine (Rapydan)
LA solutions for injection are sometimes mixed with vasoconstrictors (combination drug) to increase the duration of local anesthesia by constricting the blood vessels, thereby safely concentrating the anesthetic agent for an extended duration, as well as reducing hemorrhage.[57] Because the vasoconstrictor temporarily reduces the rate at which the systemic circulation removes the local anesthetic from the area of the injection, the maximum doses of LAs when combined with a vasoconstrictor is higher compared to the same LA without any vasoconstrictor. Occasionally, cocaine is administered for this purpose. Examples include:
- Prilocaine hydrochloride and epinephrine (trade name Citanest Forte)
- Lidocaine, bupivacaine, and epinephrine (recommended final concentrations of 0.5, 0.25, and 0.5%, respectively)
- Iontocaine, consisting of lidocaine and epinephrine
- Septocaine (trade name Septodont), a combination of articaine and epinephrine
One combination product of this type is used topically for surface anaesthesia, TAC (5-12% tetracaine,1/2000 (0.05%, 500 ppm, ½ per mille) adrenaline, 4 or 10% cocaine).
Using LA with vasoconstrictor is safe in regions supplied by end arteries.The commonly held belief that LA with vasoconstrictor can cause necrosis in extremities such as the nose, ears, fingers, and toes (due to constriction of end arteries), is invalidated, since no case of necrosis has been reported since the introduction of commercial lidocaine with epinephrine in 1948.[58]
Ester group[edit]
- Dimethocaine (Larocaine)
- Procaine (Novocaine)
- Tetracaine (Amethocaine)
Amide group[edit]
- Cinchocaine (Dibucaine)
- Lidocaine (Lignocaine)
Naturally derived[edit]
Naturally occurring local anesthetics not derived from cocaine are usually neurotoxins, and have the suffix -toxin in their names. [1] Unlike cocaine produced local anesthetics which are intracellular in effect, saxitoxin, neosaxitoxin & tetrodotoxin bind to the extracellular side of sodium channels.
History[edit]
In Peru, the ancient Incas are believed to have used the leaves of the coca plant as a local anaesthetic in addition to its stimulant properties.[59] It was also used for slave payment and is thought to play a role in the subsequent destruction of Incas culture when Spaniards realized the effects of chewing the coca leaves and took advantage of it.[59]Cocaine was isolated in 1860 and first used as a local anesthetic in 1884. The search for a less toxic and less addictive substitute led to the development of the aminoester local anesthetics stovaine in 1903 and procaine in 1904. Since then, several synthetic local anesthetic drugs have been developed and put into clinical use, notably lidocaine in 1943, bupivacaine in 1957, and prilocaine in 1959.
The invention of clinical use of local anaesthesia is credited to the Vienna School which included Sigmund Freud (1856-1939), Carl Koller (1857-1944) and Leopold Konigstein (1850-1942). They introduced local anaesthesia, using cocaine, through ‘self-experimation’ on their oral mucosa before introducing it to animal or human experimentation. The Vienna school first started using cocaine as local anaesthesia in ophthalmology and it was later incorporated into ophthalmologic practice. Dr. Halsted and Dr. Hall, in the United States in 1885 described an intraoral anaesthetic technique of blocking the inferior alveolar nerve and the antero-superior dental nerve using 4% cocaine.{[60]
Shortly after the first use of cocaine for topical anesthesia, blocks on peripheral nerves were described. Brachial plexus anesthesia by percutaneous injection through axillary and supraclavicular approaches was developed in the early 20th century. The search for the most effective and least traumatic approach for plexus anesthesia and peripheral nerve blocks continues to this day. In recent decades, continuous regional anesthesia using catheters and automatic pumps has evolved as a method of pain therapy.
Intravenous regional anesthesia was first described by August Bier in 1908. This technique is still in use and is remarkably safe when drugs of low systemic toxicity such as prilocaine are used.
Spinal anesthesia was first used in 1885, but not introduced into clinical practice until 1899, when August Bier subjected himself to a clinical experiment in which he observed the anesthetic effect, but also the typical side effect of postpunctural headache. Within a few years, spinal anesthesia became widely used for surgical anesthesia and was accepted as a safe and effective technique. Although atraumatic (noncutting-tip) cannulae and modern drugs are used today, the technique has otherwise changed very little over many decades.
Epidural anesthesia by a caudal approach had been known in the early 20th century, but a well-defined technique using lumbar injection was not developed until 1921, when Fidel Pagés published his article 'Anestesia Metamérica'. This technique was popularized in the 1930s and 1940s by Achile Mario Dogliotti. With the advent of thin, flexible catheters, continuous infusion and repeated injections have become possible, making epidural anesthesia still a highly successful technique. Besides its many uses for surgery, epidural anesthesia is particularly popular in obstetrics for the treatment of labor pain.
See also[edit]
References[edit]
- ^Ryan, T (2019). 'Tramadol as an adjunct to intra‐articular local anaesthetic infiltration in knee arthroscopy: a systematic review and meta‐analysis'. ANZ Journal of Surgery. doi:10.1111/ans.14920. PMID30684306.
- ^'Current world literature. Drugs in anaesthesia'. Current Opinion in Anesthesiology. 16 (4): 429–36. August 2003. doi:10.1097/00001503-200308000-00010. PMID17021493.
- ^Bodenham AR, Howell SJ (December 2009). 'General anaesthesia vs local anaesthesia: an ongoing story'. British Journal of Anaesthesia. 103 (6): 785–9. doi:10.1093/bja/aep310. PMID19918020.
- ^ abcdefTorpy JM, Lynm C, Golub RM (September 2011). 'JAMA patient page. Local anesthesia'. JAMA. 306 (12): 1395. doi:10.1001/jama.306.12.1395. PMID21954483.
- ^Brown AR, Weiss R, Greenberg C, Flatow EL, Bigliani LU (1993). 'Interscalene block for shoulder arthroscopy: comparison with general anesthesia'. Arthroscopy. 9 (3): 295–300. doi:10.1016/S0749-8063(05)80425-6. PMID8323615.
- ^ abMalamed SF (2013). Handbook of local anesthesia (6th ed.). St. Louis, Missouri: Elsevier. ISBN9780323074131. OCLC769141511.
- ^ abP., Chitre, A. (2016). Manual of local anaesthesia in dentistry. [Place of publication not identified]: Jaypee Brothers Medical P. ISBN978-9352501984. OCLC930829770.
- ^ abAl., Reader. Successful local anesthesia for restorative dentistry and endodontics. Nusstein, John., Drum, Melissa. Chicago. ISBN9780867156157. OCLC892911544.
- ^ ab'Nerve damage associated with peripheral nerve block'(PDF). Risks Associated with Your Anaesthetic. Section 12. January 2006. Retrieved 2007-10-10.
- ^ abBaart JA, Brand HS (2017-06-07). Local Anaesthesia in Dentistry. Springer. ISBN9783319437057.
- ^ abcdefgAronson, Jeffrey K. (2008-10-07). Meyler's Side Effects of Drugs Used in Anesthesia. Elsevier. ISBN9780444532701.
- ^Mulroy MF (2002). 'Systemic toxicity and cardiotoxicity from local anesthetics: incidence and preventive measures'. Regional Anesthesia and Pain Medicine. 27 (6): 556–61. doi:10.1053/rapm.2002.37127. PMID12430104.
- ^Stiles P, Prielipp RC (Spring 2009). 'Intralipid Treatment Of Bupicavaine Toxicity'. Anesthesia Patient Safety Foundation. 24 (1). Retrieved 12 June 2013.
- ^ abDolan R (2003-10-17). Facial plastic, reconstructive, and trauma surgery. Marcel Dekker. ISBN978-0-8247-4595-0.
- ^ abUniv. of Wisconsin, Local Anesthesia and Regional Anesthetics
- ^ abWeinberg GL, VadeBoncouer T, Ramaraju GA, Garcia-Amaro MF, Cwik MJ (April 1998). 'Pretreatment or resuscitation with a lipid infusion shifts the dose-response to bupivacaine-induced asystole in rats'. Anesthesiology. 88 (4): 1071–5. doi:10.1097/00000542-199804000-00028. PMID9579517.
- ^ abWeinberg G, Ripper R, Feinstein DL, Hoffman W (2003). 'Lipid emulsion infusion rescues dogs from bupivacaine-induced cardiac toxicity'. Regional Anesthesia and Pain Medicine. 28 (3): 198–202. doi:10.1053/rapm.2003.50041. PMID12772136.
- ^ abPicard J, Meek T (February 2006). 'Lipid emulsion to treat overdose of local anaesthetic: the gift of the glob'. Anaesthesia. 61 (2): 107–9. doi:10.1111/j.1365-2044.2005.04494.x. PMID16430560.
- ^ abRosenblatt MA, Abel M, Fischer GW, Itzkovich CJ, Eisenkraft JB (July 2006). 'Successful use of a 20% lipid emulsion to resuscitate a patient after a presumed bupivacaine-related cardiac arrest'. Anesthesiology. 105 (1): 217–8. doi:10.1097/00000542-200607000-00033. PMID16810015.
- ^ abLitz RJ, Popp M, Stehr SN, Koch T (August 2006). 'Successful resuscitation of a patient with ropivacaine-induced asystole after axillary plexus block using lipid infusion'. Anaesthesia. 61 (8): 800–1. doi:10.1111/j.1365-2044.2006.04740.x. PMID16867094.
- ^Cave G, Harvey M (September 2009). 'Intravenous lipid emulsion as antidote beyond local anesthetic toxicity: a systematic review'. Academic Emergency Medicine. 16 (9): 815–24. doi:10.1111/j.1553-2712.2009.00499.x. PMID19845549.
- ^Association of Anesthesists of Great Britain and Ireland home page
- ^Sirianni AJ, Osterhoudt KC, Calello DP, Muller AA, Waterhouse MR, Goodkin MB, Weinberg GL, Henretig FM (April 2008). 'Use of lipid emulsion in the resuscitation of a patient with prolonged cardiovascular collapse after overdose of bupropion and lamotrigine'. Annals of Emergency Medicine. 51 (4): 412–5, 415.e1. doi:10.1016/j.annemergmed.2007.06.004. PMID17766009.
- ^Home-made Lipid Rescue Kit
- ^Harvey M, Cave G (February 2007). 'Intralipid outperforms sodium bicarbonate in a rabbit model of clomipramine toxicity'. Annals of Emergency Medicine. 49 (2): 178–85, 185.e1–4. doi:10.1016/j.annemergmed.2006.07.016. PMID17098328.
- ^Harvey M, Cave G, Hoggett K (February 2009). 'Correlation of plasma and peritoneal diasylate clomipramine concentration with hemodynamic recovery after intralipid infusion in rabbits'. Academic Emergency Medicine. 16 (2): 151–6. doi:10.1111/j.1553-2712.2008.00313.x. PMID19133855.
- ^Crandell DE, Weinberg GL (April 2009). 'Moxidectin toxicosis in a puppy successfully treated with intravenous lipids'. Journal of Veterinary Emergency and Critical Care. 19 (2): 181–6. doi:10.1111/j.1476-4431.2009.00402.x. PMID19691569.
- ^Sandner-Kiesling A, Li X, Eisenach JC (December 2001). 'Morphine-induced spinal release of adenosine is reduced in neuropathic rats'. Anesthesiology. 95 (6): 1455–9. doi:10.1097/00000542-200112000-00026. PMID11748405.
- ^'BestBets: The Effect of Warming Local Anaesthetics on Pain of Infiltration'.
- ^Kampe S, Warm M, Kasper SM, Diefenbach C (July 2003). 'Concept for postoperative analgesia after pedicled TRAM flaps: continuous wound instillation with 0.2% ropivacaine via multilumen catheters. A report of two cases'. British Journal of Plastic Surgery. 56 (5): 478–83. doi:10.1016/S0007-1226(03)00180-2. PMID12890461.
- ^Meechan JG (December 1992). 'Intraligamentary anaesthesia'. Journal of Dentistry. 20 (6): 325–32. doi:10.1016/0300-5712(92)90018-8. PMID1452871.
- ^Blanton PL, Jeske AH (June 2003). 'The key to profound local anesthesia: neuroanatomy'. Journal of the American Dental Association. 134 (6): 753–60. doi:10.14219/jada.archive.2003.0262. PMID12839412.
- ^Boynes SG (1 June 2018 ). 'Intraligamentary Injections in Dentistry'(PDF). Dental Academy of Continuing Education.Check date values in:
|date=(help) - ^Meechan JG (November 2002). 'Supplementary routes to local anaesthesia'. International Endodontic Journal. 35 (11): 885–96. doi:10.1046/j.1365-2591.2002.00592.x. PMID12453016.
- ^D'Souza JE, Walton RE, Peterson LC (March 1987). 'Periodontal ligament injection: an evaluation of the extent of anesthesia and postinjection discomfort'. Journal of the American Dental Association. 114 (3): 341–4. doi:10.14219/jada.archive.1987.0080. PMID3470356.
- ^Shastry SP, Kaul R, Baroudi K, Umar D (December 2014). 'Hemophilia A: Dental considerations and management'. Journal of International Society of Preventive & Community Dentistry. 4 (Suppl 3): S147–52. doi:10.4103/2231-0762.149022. PMC4304051. PMID25625071.
- ^Nazif M (January 1970). 'Local anesthesia for patients with hemophilia'. ASDC Journal of Dentistry for Children. 37 (1): 79–84. PMID4904493.
- ^Moore PA, Haas DA (October 2010). 'Paresthesias in dentistry'. Dental Clinics of North America. 54 (4): 715–30. doi:10.1016/j.cden.2010.06.016. PMID20831934.
- ^ abShabazfar N, Daubländer M, Al-Nawas B, Kämmerer PW (2014). 'Periodontal intraligament injection as alternative to inferior alveolar nerve block--meta-analysis of the literature from 1979 to 2012'. Clinical Oral Investigations. 18 (2): 351–8. doi:10.1007/s00784-013-1113-1. PMID24077785.
- ^Nelson PW. Injection system. J Am Dent Assoc. 1981.
- ^Galili D, Kaufman E, Garfunkel AD, et al. Intraligamental anesthesia – a histological study. International Journal of Oral and Maxillofacial Surgery. 1984.
- ^Pashley D (October 1986). 'Systemic effects of intraligamental injections'. Journal of Endodontics. 12 (10): 501–4. doi:10.1016/s0099-2399(86)80206-0. PMID3465856.
- ^Walton RE, Abbott BJ (October 1981). 'Periodontal ligament injection: a clinical evaluation'. Journal of the American Dental Association. 103 (4): 571–5. doi:10.14219/jada.archive.1981.0307. PMID6945341.
- ^Smith GN, Walton RE, Abbott BJ (December 1983). 'Clinical evaluation of periodontal ligament anesthesia using a pressure syringe'. Journal of the American Dental Association. 107 (6): 953–6. doi:10.14219/jada.archive.1983.0357. PMID6581222.
- ^ abRoberts GJ, Holzel HS, Sury MR, Simmons NA, Gardner P, Longhurst P (January 1997). 'Dental bacteremia in children'. Pediatric Cardiology. 18 (1): 24–7. doi:10.1007/s002469900103. PMID8960488.
- ^Kaufman E, Galili D, Garfunkel AA (March 1983). 'Intraligamentary anesthesia: a clinical study'. The Journal of Prosthetic Dentistry. 49 (3): 337–9. doi:10.1016/0022-3913(83)90273-1. PMID6573480.
- ^Malamed SF (February 1982). 'The periodontal ligament (PDL) injection: an alternative to inferior alveolar nerve block'. Oral Surgery, Oral Medicine, and Oral Pathology. 53 (2): 117–21. doi:10.1016/0030-4220(82)90273-0. PMID6949113.
- ^ abMeechan JG (January 1999). 'How to overcome failed local anaesthesia'. British Dental Journal. 186 (1): 15–20. doi:10.1038/sj.bdj.4800006. PMID10028738.
- ^Walton RE, Garnick JJ (January 1982). 'The periodontal ligament injection: histologic effects on the periodontium in monkeys'. Journal of Endodontics. 8 (1): 22–6. doi:10.1016/S0099-2399(82)80312-9. PMID6948904.
- ^Hochman MN, Friedman MJ, Williams W, Hochman CB (June 2006). 'Interstitial tissue pressure associated with dental injections: a clinical study'. Quintessence International. 37 (6): 469–76. PMID16752703.
- ^Aggarwal V, Singla M, Miglani S, Kohli S, Sharma V, Bhasin SS (January 2018). 'Does the volume of supplemental intraligamentary injections affect the anaesthetic success rate after a failed primary inferior alveolar nerve block? A randomized-double blind clinical trial'. International Endodontic Journal. 51 (1): 5–11. doi:10.1111/iej.12773. PMID28370327.
- ^Berlin J, Nusstein J, Reader A, Beck M, Weaver J (March 2005). 'Efficacy of articaine and lidocaine in a primary intraligamentary injection administered with a computer-controlled local anesthetic delivery system'. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontics. 99 (3): 361–6. doi:10.1016/j.tripleo.2004.11.009. PMID15716846.
- ^Froum SJ, Tarnow D, Caiazzo A, Hochman MN (September 2000). 'Histologic response to intraligament injections using a computerized local anesthetic delivery system. A pilot study in mini-swine'. Journal of Periodontology. 71 (9): 1453–9. doi:10.1902/jop.2000.71.9.1453. PMID11022775.
- ^ abcdGow-Gates GA (April 1998). 'The Gow-Gates mandibular block: regional anatomy and analgesia'. Australian Endodontic Journal. 24 (1): 18–9. doi:10.1111/j.1747-4477.1998.tb00251.x. PMID11431805.
- ^'Allergic Reactions'. Cleveland Clinic. Retrieved 11 April 2014.
- ^Arnold Stern (2002). Pharmacology: PreTest self-assessment and review. New York: McGraw-Hill, Medical Pub. Division. ISBN978-0-07-136704-2.
- ^Yagiela JA (1995). 'Vasoconstrictor agents for local anesthesia'. Anesthesia Progress. 42 (3–4): 116–20. PMC2148913. PMID8934977.
- ^Nielsen LJ, Lumholt P, Hölmich LR (October 2014). '[Local anaesthesia with vasoconstrictor is safe to use in areas with end-arteries in fingers, toes, noses and ears]'. Ugeskrift for Laeger. 176 (44): 44. PMID25354008.
- ^ ab'Cocaine's use: From the Incas to the U.S.'Boca Raton News. 4 April 1985. Retrieved 2 February 2014.
- ^López-Valverde A, de Vicente J, Martínez-Domínguez L, de Diego RG (July 2014). 'Local anaesthesia through the action of cocaine, the oral mucosa and the Vienna group'. British Dental Journal. 217 (1): 41–3. doi:10.1038/sj.bdj.2014.546. PMID25012333.
External links[edit]
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Local_anesthetic&oldid=904185785'
- Blog
- Home
- New Page
- Gta San Andreas Download Full Game
- Windows 7 Premium Product Key Generator
- Doom Project Brutality 3.0 Download
- Woody Woodpecker Tomcat Combat
- Witcher 3 Graphic Mods
- Solidworks Free Download 2019 Free
- Arma 3 Breaking Point Chernarus Map
- Gta 5 Bodybuilder Mod
- Windows Live Calendar Update Error
- Witcher 3 Crashes To Desktop
- Baixar Avast Premier 2019 Crackeado
- Battle In The Forgotten City
- Space Battle Of Scarif
- War Is Hell Game
- Dnc Software For Fanuc Free Download
- Sim Settlements Far Harbor
- Focus Mk2 Remove Glovebox
- Install Hp Psc 950 On Windows 10
- Dbc File Viewer Online
- Chemical Structure Of Local Anaesthetics
- Pramukh Gujarati Font Converter
- A Deep Commitment To Purple
